New developments in atrial antiarrhythmic drug therapy
- PMID: 20179721
- PMCID: PMC2844858
- DOI: 10.1038/nrcardio.2009.245
New developments in atrial antiarrhythmic drug therapy
Abstract
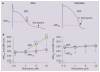
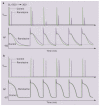
Atrial fibrillation (AF) is a growing clinical problem associated with increased morbidity and mortality. Currently available antiarrhythmic drugs (AADs), although highly effective in acute cardioversion of paroxysmal AF, are generally only moderately successful in long-term maintenance of sinus rhythm. The use of AADs is often associated with an increased risk of ventricular proarrhythmia, extracardiac toxicity, and exacerbation of concomitant diseases such as heart failure. AF is commonly associated with intracardiac and extracardiac disease, which can modulate the efficacy and safety of AAD therapy. In light of the multifactorial intracardiac and extracardiac causes of AF generation, current development of anti-AF agents is focused on modulation of ion channel activity as well as on upstream therapies that reduce structural substrates. The available data indicate that multiple ion channel blockers exhibiting potent inhibition of peak I(Na) with relatively rapid unbinding kinetics, as well as inhibition of late I(Na) and I(Kr), may be preferable for the management of AF when considering both safety and efficacy.
Figures


Similar articles
-
New pharmacological targets and treatments for atrial fibrillation.Trends Pharmacol Sci. 2010 Aug;31(8):364-71. doi: 10.1016/j.tips.2010.05.001. Epub 2010 May 31. Trends Pharmacol Sci. 2010. PMID: 20605645 Review.
-
Novel anti-arrhythmic drugs for atrial fibrillation management.Curr Vasc Pharmacol. 2007 Jul;5(3):185-95. doi: 10.2174/157016107781024073. Curr Vasc Pharmacol. 2007. PMID: 17627562 Review.
-
Antiarrhythmic therapy in atrial fibrillation.Pharmacol Ther. 2010 Oct;128(1):129-45. doi: 10.1016/j.pharmthera.2010.06.004. Epub 2010 Jul 17. Pharmacol Ther. 2010. PMID: 20624425 Review.
-
Current antiarrhythmic drugs: an overview of mechanisms of action and potential clinical utility.J Cardiovasc Electrophysiol. 1999 Feb;10(2):283-301. doi: 10.1111/j.1540-8167.1999.tb00674.x. J Cardiovasc Electrophysiol. 1999. PMID: 10090235 Review.
-
The Past, Present, and Potential Future of Sodium Channel Block as an Atrial Fibrillation Suppressing Strategy.J Cardiovasc Pharmacol. 2015 Nov;66(5):432-40. doi: 10.1097/FJC.0000000000000271. J Cardiovasc Pharmacol. 2015. PMID: 25923324
Cited by
-
Advances in the Pharmacologic Management of Atrial Fibrillation.Card Electrophysiol Clin. 2011 Mar;3(1):157-167. doi: 10.1016/j.ccep.2010.10.006. Card Electrophysiol Clin. 2011. PMID: 21731596 Free PMC article. No abstract available.
-
Atrial-selective sodium channel block by dronedarone: sufficient to terminate atrial fibrillation?Naunyn Schmiedebergs Arch Pharmacol. 2011 Aug;384(2):109-14. doi: 10.1007/s00210-011-0647-5. Epub 2011 May 4. Naunyn Schmiedebergs Arch Pharmacol. 2011. PMID: 21541758 No abstract available.
-
Advances in the Pharmacological Treatment of Atrial Fibrillation.Curr Med Lit Cardiol. 2010 Jan 1;29(1):1-5. Curr Med Lit Cardiol. 2010. PMID: 21152111 Free PMC article. No abstract available.
-
Potassium currents in the heart: functional roles in repolarization, arrhythmia and therapeutics.J Physiol. 2017 Apr 1;595(7):2229-2252. doi: 10.1113/JP272883. Epub 2017 Jan 5. J Physiol. 2017. PMID: 27808412 Free PMC article. Review.
-
German Cardiac Society Working Group on Cellular Electrophysiology state-of-the-art paper: impact of molecular mechanisms on clinical arrhythmia management.Clin Res Cardiol. 2019 Jun;108(6):577-599. doi: 10.1007/s00392-018-1377-1. Epub 2018 Oct 10. Clin Res Cardiol. 2019. PMID: 30306295 Review.
References
-
- Fuster V, et al. ACC/AHA/ESC 2006 guidelines for the management of patients with atrial fibrillation--executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Revise the 2001 Guidelines for the Management of Patients With Atrial Fibrillation) J. Am. Coll. Cardiol. 2006;48:854–906. - PubMed
-
- Naccarelli GV, Gonzalez MD. Atrial fibrillation and the expanding role of catheter ablation: do antiarrhythmic drugs have a future? J. Cardiovasc. Pharmacol. 2008;52:203–209. - PubMed
-
- Roux JF, et al. Antiarrhythmics after ablation of atrial aibrillation (5A Study) Circulation. 2009;120:1036–1040. - PubMed
-
- Cain ME, Curtis AB. Rhythm control in atrial fibrillation—one setback after another. N. Engl. J. Med. 2008;358:2725–2727. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

