Interactions between age, stress and insulin on cognition: implications for Alzheimer's disease
- PMID: 20182419
- PMCID: PMC3055481
- DOI: 10.1038/npp.2010.13
Interactions between age, stress and insulin on cognition: implications for Alzheimer's disease
Abstract
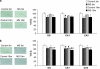
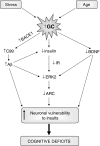
There is much interest in understanding the mechanisms responsible for interactions among stress, aging, memory and Alzheimer's disease. Glucocorticoid secretion associated with early life stress may contribute to the variability of the aging process and to the development of neuro- and psychopathologies. Maternal separation (MS), a model of early life stress in which rats experience 3 h of daily separation from the dam during the first 3 weeks of life, was used to study the interactions between stress and aging. Young (3 months) MS rats showed an altered hypothalamic-pituitary-adrenal (HPA) axis reactivity, depressive-like behavior in the Porsolt swimming test and cognitive impairments in the Morris water maze and new object recognition test that persisted in aged (18 months) rats. Levels of insulin receptor, phosphorylated insulin receptor and markers of downstream signaling pathways (pAkt, pGSK3 beta, pTau, and pERK1 levels) were significantly decreased in aged rats. There was a significant decrease in pERK2 and in the plasticity marker ARC in MS aged rats compared with single MS or aged rats. It is interesting to note that there was a significant increase in the C99 : C83 ratio, A beta levels, and BACE1 levels the hippocampus of MS aged rats, suggesting that in aged rats subjected to early life stress, there was an increase in the amyloidogenic processing of amyloid precursor protein (APP). These results are integrated in a tentative mechanism through which aging interplay with stress to influence cognition as the basis of Alzheimer disease (AD). The present results may provide the proof-of-concept for the use of glucocorticoid-/insulin-related drugs in the treatment of AD.
Figures
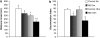

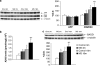
Comment in
-
Why do we need to use animal models to study cognition and aging?Neuropsychopharmacology. 2010 Jul;35(8):1621-2. doi: 10.1038/npp.2010.45. Neuropsychopharmacology. 2010. PMID: 20551899 Free PMC article. No abstract available.
Similar articles
-
Cognitive impairment associated to HPA axis hyperactivity after maternal separation in rats.Psychoneuroendocrinology. 2007 Apr;32(3):256-66. doi: 10.1016/j.psyneuen.2006.12.013. Epub 2007 Feb 20. Psychoneuroendocrinology. 2007. PMID: 17307298
-
Stress contributes to the development of central insulin resistance during aging: implications for Alzheimer's disease.Biochim Biophys Acta. 2013 Dec;1832(12):2332-9. doi: 10.1016/j.bbadis.2013.09.013. Epub 2013 Oct 1. Biochim Biophys Acta. 2013. PMID: 24090692
-
Effects of early maternal separation on biobehavioral and neuropathological markers of Alzheimer's disease in adult male rats.Curr Alzheimer Res. 2013 May 1;10(4):420-32. doi: 10.2174/1567205011310040007. Curr Alzheimer Res. 2013. PMID: 23305081
-
Effects of stress and stress hormones on amyloid-beta protein and plaque deposition.J Alzheimers Dis. 2009;18(2):459-69. doi: 10.3233/JAD-2009-1152. J Alzheimers Dis. 2009. PMID: 19584430 Free PMC article. Review.
-
Adverse stress, hippocampal networks, and Alzheimer's disease.Neuromolecular Med. 2010 Mar;12(1):56-70. doi: 10.1007/s12017-009-8107-9. Epub 2009 Nov 27. Neuromolecular Med. 2010. PMID: 19943124 Free PMC article. Review.
Cited by
-
Stress-Induced Neurodegeneration: The Potential for Coping as Neuroprotective Therapy.Am J Alzheimers Dis Other Demen. 2020 Jan-Dec;35:1533317520960873. doi: 10.1177/1533317520960873. Am J Alzheimers Dis Other Demen. 2020. PMID: 32969239 Free PMC article.
-
Insulin can induce the expression of a memory-related synaptic protein through facilitating AMPA receptor endocytosis in rat cortical neurons.Cell Mol Life Sci. 2014 Oct;71(20):4069-80. doi: 10.1007/s00018-014-1620-5. Epub 2014 Apr 6. Cell Mol Life Sci. 2014. PMID: 24705985 Free PMC article.
-
The hypothalamus in Alzheimer's disease: a Golgi and electron microscope study.Am J Alzheimers Dis Other Demen. 2015 Aug;30(5):478-87. doi: 10.1177/1533317514556876. Epub 2014 Nov 7. Am J Alzheimers Dis Other Demen. 2015. PMID: 25380804 Free PMC article.
-
APOE4 genotype exacerbates the depression-like behavior of mice during aging through ATP decline.Transl Psychiatry. 2021 Oct 5;11(1):507. doi: 10.1038/s41398-021-01631-0. Transl Psychiatry. 2021. PMID: 34611141 Free PMC article.
-
Progesterone exerts antidepressant-like effect in a mouse model of maternal separation stress through mitigation of neuroinflammatory response and oxidative stress.Pharm Biol. 2020 Jan 1;58(1):64-71. doi: 10.1080/13880209.2019.1702704. Pharm Biol. 2020. PMID: 31873049 Free PMC article.
References
-
- Aisa B, Tordera R, Lasheras B, Del Río J, Ramírez MJ. Cognitive impairment associated to HPA axis hyperactivity after maternal separation in rats. Psychoneuroendocrinology. 2007;32:256–266. - PubMed
-
- Amatruda JM, Livingston JN, Lockwood DH. Cellular mechanisms in selected states of insulin resistance: human obesity, glucocorticoid excess, and chronic renal failure. Diabetes Metab Rev. 1985;1:293–317. - PubMed
-
- Berger-Sweeney J, McPhie DL, Arters JA, Greenan J, Oster-Granite ML, Neve RL. Impairments in learning and memory accompanied by neurodegeneration in mice transgenic for the carboxyl-terminus of the amyloid precursor protein. Mol Brain Res. 1999;66:150–162. - PubMed
-
- Berton O, Nestler EJ. New approaches to antidepressant drug discovery: beyond monoamines. Nat Rev Neurosci. 2006;7:137–151. - PubMed
-
- Born J, Ditschuneit I, Schreiber M, Dodt C, Fehm HL. Effects of age and gender on pituitary-adrenocortical responsiveness in humans. Eur J Endocrinol. 1995;132:705–711. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

