Oral insulin: a comparison with subcutaneous regular human insulin in patients with type 2 diabetes
- PMID: 20185734
- PMCID: PMC2875439
- DOI: 10.2337/dc09-1807
Oral insulin: a comparison with subcutaneous regular human insulin in patients with type 2 diabetes
Abstract
Objective: To determine the pharmacokinetic and pharmacodynamic properties of an oral insulin (OI) formulation compared with subcutaneously injected regular human insulin (RHI).
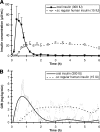
Research design and methods: Ten male patients with type 2 diabetes (means +/- SD; A1C 7.0 +/- 1.1%; BMI 28.3 +/- 2.7 kg/m(2)) received either 300 units of insulin combined with 400 mg of delivery agent orally or 15 units RHI subcutaneously under isoglycemic clamp conditions.
Results: Maximum insulin concentration was greater and onset of action was faster with OI (C(max) 93 +/- 71 vs. 33 +/- 11 microU/ml; AUC(GIR)((0-1h)) 173 +/- 86 vs. 27 +/- 32 mg/kg; P < 0.05). Mean insulin concentration and glucose infusion rate returned to baseline within 3 h after OI administration. Relative bioavailability of OI was 7 +/- 4% (1st 2 h).
Conclusions: This proof-of-concept study demonstrated that absorption of OI is feasible under fasting conditions. OI has a fast onset and a short duration of action but also shows a rather high between-subject variability in absorption.
Trial registration: ClinicalTrials.gov NCT00982254.
Figures
References
-
- Wajcberg E, Miyazaki Y, Triplitt C, Cersosimo E, DeFronzo RA: Dose-response effect of a single administration of oral hexyl-insulin monoconjugate 2 in healthy nondiabetic subjects. Diabetes Care 2004;27:2868–2873 - PubMed
-
- Blackard WG, Nelson NC: Portal and peripheral vein immunoreactive insulin concentrations before and after glucose infusion. Diabetes 1970;19:302–306 - PubMed
-
- Yadav N, Morris G, Harding SE, Ang S, Adams GG: Various non-injectable delivery systems for the treatment of diabetes mellitus. Endocr Metab Immune Disord Drug Targets 2009;9:1–13 - PubMed
-
- Lassmann-Vague V, Raccah D: Alternatives routes of insulin delivery. Diabete Metab 2006;32:513–522 - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

