A novel effect of growth hormone on macrophage modulates macrophage-dependent adipocyte differentiation
- PMID: 20185763
- PMCID: PMC2869256
- DOI: 10.1210/en.2009-1194
A novel effect of growth hormone on macrophage modulates macrophage-dependent adipocyte differentiation
Abstract
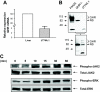
The GH receptor (GHR) is expressed on macrophages. However, the precise role of GH in regulation of macrophage function is unclear. We hypothesized that soluble factors including cytokines produced by macrophages in a GH-dependent manner regulate adipogenesis. We confirmed expression and functional integrity of the GHR in the J774A.1 macrophage cells. Conditioned medium (CM) from macrophages inhibited adipogenesis in a 3T3-L1 adipogenesis assay. CM from GH-treated macrophages decreased the inhibitory effect of CM from macrophages on adipogenesis. This effect on preadipocyte differentiation was active only during the first (early) phase of adipocyte differentiation. CM from stromal vascular compartment macrophages of mice with macrophage-specific deletion of the GHR exhibited more inhibitory effect on 3T3-L1 preadipocyte differentiation compared with CM from stromal vascular compartment macrophages of control mice, indicating that intact GH action in primary macrophages also increases preadipocyte differentiation. GH did not increase IGF-1 expression in macrophages. PCR array analysis identified IL-1beta as a candidate cytokine whose expression was altered by GH in macrophages. Levels of IL-1beta mRNA and protein were significantly decreased in GH-treated J774A.1 macrophages. Nuclear factor-kappaB stimulates IL-1beta gene expression, and GH induced a significant decrease in the levels of phosphorylated nuclear factor-kappaB in macrophages. IL-1beta is a known inhibitor of adipogenesis, and these results support GH-dependent down-regulation of macrophage IL-1beta expression as one mechanism for the observed increase in adipogenesis with CM from GH-treated macrophages. We conclude that GH decreases secretion of IL-1beta by the macrophage and thus in a paracrine manner increases adipocyte differentiation. These results provide a novel mechanism for GH's actions in the control of adipogenesis.
Figures



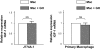


Similar articles
-
New insights of growth hormone (GH) actions from tissue-specific GH receptor knockouts in mice.Arch Endocrinol Metab. 2019 Nov-Dec;63(6):557-567. doi: 10.20945/2359-3997000000185. Arch Endocrinol Metab. 2019. PMID: 31939480 Free PMC article. Review.
-
Growth hormone and prolactin receptors in adipogenesis: STAT-5 activation, suppressors of cytokine signaling, and regulation of insulin-like growth factor I.Horm Res. 2006;66(3):101-10. doi: 10.1159/000093667. Epub 2006 May 30. Horm Res. 2006. PMID: 16735796
-
The activation state of macrophages alters their ability to suppress preadipocyte apoptosis.J Endocrinol. 2012 Jul;214(1):21-9. doi: 10.1530/JOE-12-0114. Epub 2012 May 3. J Endocrinol. 2012. PMID: 22556272
-
α-Naphthoflavone modulates inflammatory response in adipocytes-macrophages interaction through NFκB signaling.Int J Clin Exp Pathol. 2014 Oct 15;7(11):7768-74. eCollection 2014. Int J Clin Exp Pathol. 2014. PMID: 25550814 Free PMC article.
-
The Modulatory Role of Growth Hormone in Inflammation and Macrophage Activation.Endocrinology. 2022 Aug 1;163(8):bqac088. doi: 10.1210/endocr/bqac088. Endocrinology. 2022. PMID: 35695371 Review.
Cited by
-
New insights of growth hormone (GH) actions from tissue-specific GH receptor knockouts in mice.Arch Endocrinol Metab. 2019 Nov-Dec;63(6):557-567. doi: 10.20945/2359-3997000000185. Arch Endocrinol Metab. 2019. PMID: 31939480 Free PMC article. Review.
-
Endocrine parameters and phenotypes of the growth hormone receptor gene disrupted (GHR-/-) mouse.Endocr Rev. 2011 Jun;32(3):356-86. doi: 10.1210/er.2010-0009. Epub 2010 Dec 1. Endocr Rev. 2011. PMID: 21123740 Free PMC article. Review.
-
Circulating sex steroids coregulate adipose tissue immune cell populations in healthy men.Am J Physiol Endocrinol Metab. 2017 Nov 1;313(5):E528-E539. doi: 10.1152/ajpendo.00075.2017. Epub 2017 Jul 11. Am J Physiol Endocrinol Metab. 2017. PMID: 28698282 Free PMC article.
-
Interleukin-1 beta: a potential link between stress and the development of visceral obesity.BMC Physiol. 2012 Jun 27;12:8. doi: 10.1186/1472-6793-12-8. BMC Physiol. 2012. PMID: 22738239 Free PMC article. Review.
-
Removal of growth hormone receptor (GHR) in muscle of male mice replicates some of the health benefits seen in global GHR-/- mice.Aging (Albany NY). 2015 Jul;7(7):500-12. doi: 10.18632/aging.100766. Aging (Albany NY). 2015. PMID: 26233957 Free PMC article.
References
-
- Møller N, Jørgensen JO 2009 Effects of growth hormone on glucose, lipid, and protein metabolism in human subjects. Endocr Rev 30:152–177 - PubMed
-
- Smith JR, Benghuzzi H, Tucci M, Puckett A, Hughes JL 2000 The effects of growth hormone and insulin-like growth factor on the proliferation rate and morphology of RAW 264.7 macrophages. Biomed Sci Instrum 36:111–116 - PubMed
-
- Warwick-Davies J, Lowrie DB, Cole PJ 1995 Growth hormone is a human macrophage activating factor. Priming of human monocytes for enhanced release of H2O2. J Immunol 154:1909–1918 - PubMed
-
- Hochberg Z, Hertz P, Maor G, Oiknine J, Aviram M 1992 Growth hormone and insulin-like growth factor-I increase macrophage uptake and degradation of low density lipoprotein. Endocrinology 131:430–435 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

