Quantitative analysis of adhesive resin in the hybrid layer using Raman spectroscopy
- PMID: 20186729
- PMCID: PMC2875550
- DOI: 10.1002/jbm.a.32692
Quantitative analysis of adhesive resin in the hybrid layer using Raman spectroscopy
Abstract
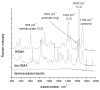
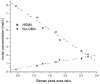
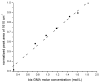
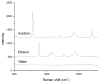
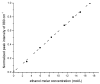
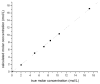
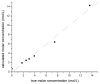
The objective was to determine absolute molar concentration of adhesive resin components in the hybrid layer by establishing methods based on Raman spectroscopy fundamentals. The hybrid layer was treated as a three-component system consisting of collagen and an adhesive resin containing two monomers. Adhesive standard specimens and Raman peak area ratios obtained with a 785 nm excitation wavelength were used to construct separate calibration curves for comonomer relative molar concentration and Bis-GMA absolute molar concentration. As collagen and water had no measurable peaks in the fingerprint region, a dilution coefficient K(j) was defined to describe their impact on Raman peak area and to calculate HEMA absolute molar concentration. Methodology was validated using an analogous system containing acetone/ethanol/water. The absolute molar concentration of Bis-GMA and HEMA decreased 87% and 83%, respectively, from the top quarter to the middle of the hybrid layer. Additionally, less Bis-GMA penetrated the hybrid layer than HEMA, as indicated by the approximately 20% decrease in comonomer molar concentration ratio between the adhesive resin layer and the top half of the hybrid layer. Lack of complete monomer infiltration will further challenge dentin-adhesive bond longevity. (c) 2010 Wiley Periodicals, Inc. J Biomed Mater Res, 2010.
Figures


Similar articles
-
In vitro enzymatic biodegradation of adhesive resin in the hybrid layer.J Biomed Mater Res A. 2010 Jul;94(1):187-92. doi: 10.1002/jbm.a.32689. J Biomed Mater Res A. 2010. PMID: 20127999 Free PMC article.
-
Mapping water and monomer gradients in the adhesive/dentin interface with confocal micro-Raman imaging.Dent Mater. 2025 Apr;41(4):473-481. doi: 10.1016/j.dental.2025.02.002. Epub 2025 Feb 17. Dent Mater. 2025. PMID: 39966031
-
Ethanol-wet bonding and chlorhexidine improve resin-dentin bond durability: quantitative analysis using raman spectroscopy.J Adhes Dent. 2014 Oct;16(5):441-50. doi: 10.3290/j.jad.a32695. J Adhes Dent. 2014. PMID: 25202747
-
Use of micro-Raman spectroscopy to investigate hybrid layer quality in demineralized root dentine.J Biomed Mater Res B Appl Biomater. 2010 Oct;95(1):62-8. doi: 10.1002/jbm.b.31683. J Biomed Mater Res B Appl Biomater. 2010. PMID: 20690175
-
Morphological and chemical characterization of bonding hydrophobic adhesive to dentin using ethanol wet bonding technique.Dent Mater. 2009 Aug;25(8):1050-7. doi: 10.1016/j.dental.2009.03.006. Epub 2009 Apr 16. Dent Mater. 2009. PMID: 19371945 Free PMC article.
Cited by
-
Scale dependent nanomechanical properties of dentin adhesive and adhesive-collagen composite.Front Dent Med. 2024 Nov 28;5:1423461. doi: 10.3389/fdmed.2024.1423461. eCollection 2024. Front Dent Med. 2024. PMID: 39917649 Free PMC article.
-
In vitro bioactivity of newly introduced dual-cured resin-modified calcium silicate cement.Dent Res J (Isfahan). 2022 Jan 28;19:1. doi: 10.4103/1735-3327.336686. eCollection 2022. Dent Res J (Isfahan). 2022. PMID: 35308449 Free PMC article.
-
A Practical Primer: Raman Spectroscopy for Monitoring of Photopolymerization Systems.Polymers (Basel). 2023 Sep 20;15(18):3835. doi: 10.3390/polym15183835. Polymers (Basel). 2023. PMID: 37765689 Free PMC article. Review.
-
Swelling equilibrium of dentin adhesive polymers formed on the water-adhesive phase boundary: experiments and micromechanical model.Acta Biomater. 2014 Jan;10(1):330-42. doi: 10.1016/j.actbio.2013.09.017. Epub 2013 Sep 26. Acta Biomater. 2014. PMID: 24076070 Free PMC article.
-
Structure-property relationships for wet dentin adhesive polymers.Biointerphases. 2018 Dec 17;13(6):061004. doi: 10.1116/1.5058072. Biointerphases. 2018. PMID: 30558430 Free PMC article.
References
-
- Nakabayashi N, Kojima K, Masuhara E. The promotion of adhesion by the infiltration of monomers into tooth substrates. Shika Zairyo Kikai. 1982;1:78–81. - PubMed
-
- Armstrong SR, Keller JC, Boyer DB. Mode of Failure in the Dentin-Adhesive Resin-Resin Composite Bonded Joint as Determined by Strength-Based (μTBS) and Fracture-based (CNSB) Mechanical Testing. Dent Mater. 2000;17:201–210. - PubMed
-
- Hashimoto M, De Munck J, Ito S, Sano H, Kaga M, Oguchi H, Van Meerbeek B, Pashley DH. In vitro effect of nanoleakage expression on resin-dentin bond strengths analyzed by microtensile bond test, SEM/EDX and TEM. Biomaterials. 2004;25(25):5565–74. - PubMed
-
- Ito S, Tay FR, Hashimoto M, Yoshiyama M, Saito T, Brackett WW, Waller JL, Pashley DH. Effects of multiple coatings of two all-in-one adhesives on dentin bonding. J Adhes Dent. 2005;7(2):133–141. - PubMed
-
- Koshiro K, Sidhu SK, Inoue S, Ikeda T, Sano H. New concept of resin-dentin interfacial adhesion: the nanointeraction zone. J Biomed Mater Res Part B: Applied BioMater. 2006;77B(2):401–408. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

