Immunostimulatory CpG oligonucleotides: Effect on gene expression and utility as vaccine adjuvants
- PMID: 20188247
- PMCID: PMC2830905
- DOI: 10.1016/j.vaccine.2009.10.094
Immunostimulatory CpG oligonucleotides: Effect on gene expression and utility as vaccine adjuvants
Abstract
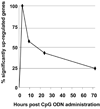
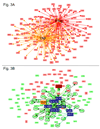
Synthetic oligodeoxynucleotides (ODN) containing unmethylated CpG motifs mimic the immunostimulatory activity of bacterial DNA. CpG ODN directly stimulate B cells and plasmacytoid dendritic cells (pDC), promote the production of Th1 and pro-inflammatory cytokines, and trigger the maturation/activation of professional antigen presenting cells. CpG ODN are finding use as vaccine adjuvants, where they increase the speed, magnitude and duration of vaccine-specific immune responses. For example, CpG ODN significantly prolong the protection induced by AVA (Anthrax Vaccine Adsorbed). Unexpectedly, a majority of animals immunized with CpG-adjuvanted AVA maintain resistance to anthrax infection even after their Ab titers decline to sub-protective levels. This survival is mediated by the de novo production of protective Abs by high affinity long-lived memory B cells. The immunostimulatory activity of CpG ODN was probed at the molecular level by microarray. Results show that a small group of 'inducers' rapidly up-regulated a large network genes following CpG treatment of mice. This stimulatory activity is quenched by 'suppressors' that down-regulate the expression of targeted genes, including most of the 'inducers'. These findings shed light on the mechanism underlying CpG-mediated immune activation and therapeutic activity.
Published by Elsevier Ltd.
Figures

Similar articles
-
Effect of CpG oligonucleotides on vaccine-induced B cell memory.J Immunol. 2008 Oct 15;181(8):5785-90. doi: 10.4049/jimmunol.181.8.5785. J Immunol. 2008. PMID: 18832738 Free PMC article.
-
CpG oligonucleotides accelerate and boost the immune response elicited by AVA, the licensed anthrax vaccine.Expert Rev Vaccines. 2006 Jun;5(3):365-9. doi: 10.1586/14760584.5.3.365. Expert Rev Vaccines. 2006. PMID: 16827620 Review.
-
CpG oligodeoxynucleotides adsorbed onto polylactide-co-glycolide microparticles improve the immunogenicity and protective activity of the licensed anthrax vaccine.Infect Immun. 2005 Feb;73(2):828-33. doi: 10.1128/IAI.73.2.828-833.2005. Infect Immun. 2005. PMID: 15664922 Free PMC article.
-
CpG oligonucleotides improve the protective immune response induced by the anthrax vaccination of rhesus macaques.Vaccine. 2004 Jul 29;22(21-22):2881-6. doi: 10.1016/j.vaccine.2003.12.020. Vaccine. 2004. PMID: 15246624
-
Clinical evaluation of CpG oligonucleotides as adjuvants for vaccines targeting infectious diseases and cancer.Vaccine. 2014 Nov 12;32(48):6377-89. doi: 10.1016/j.vaccine.2014.06.065. Epub 2014 Jun 24. Vaccine. 2014. PMID: 24975812 Free PMC article. Review.
Cited by
-
Anti-protease and immunomodulatory activities of bacteria associated with Caribbean sponges.Mar Biotechnol (NY). 2011 Oct;13(5):883-92. doi: 10.1007/s10126-010-9349-0. Epub 2011 Jan 11. Mar Biotechnol (NY). 2011. PMID: 21222136 Free PMC article.
-
DNA vaccines for targeting bacterial infections.Expert Rev Vaccines. 2010 Jul;9(7):747-63. doi: 10.1586/erv.10.57. Expert Rev Vaccines. 2010. PMID: 20624048 Free PMC article. Review.
-
A DNA prime-protein boost vaccination strategy targeting turkey coronavirus spike protein fragment containing neutralizing epitope against infectious challenge.Vet Immunol Immunopathol. 2013 Apr 15;152(3-4):359-69. doi: 10.1016/j.vetimm.2013.01.009. Epub 2013 Feb 1. Vet Immunol Immunopathol. 2013. PMID: 23428360 Free PMC article.
-
Characterization of immunogenicity of avian influenza antigens encapsulated in PLGA nanoparticles following mucosal and subcutaneous delivery in chickens.PLoS One. 2018 Nov 1;13(11):e0206324. doi: 10.1371/journal.pone.0206324. eCollection 2018. PLoS One. 2018. PMID: 30383798 Free PMC article.
-
Adjuvant-carrying synthetic vaccine particles augment the immune response to encapsulated antigen and exhibit strong local immune activation without inducing systemic cytokine release.Vaccine. 2014 May 19;32(24):2882-95. doi: 10.1016/j.vaccine.2014.02.027. Epub 2014 Mar 1. Vaccine. 2014. PMID: 24593999 Free PMC article.
References
-
- Ballas ZD, Rasmussen WL, Krieg AM. Induction of NK activity in murine and human cells by CpG motifs in oligodeoxynucleotides and bacterial DNA. J. Immunol. 1996;157:1840–1847. - PubMed
-
- Bauer M, Redecke V, Ellwart JW, Sherer B, Kremer JP, Wagner H, Lipford GB. Bacterial CpG DNA triggers activation and maturation of human CD11c(−), CD123(+) dendritic cells. J. Immunol. 2001;166:5000–5007. - PubMed
-
- Branda RF, Moore AL, Lafayette AR, Mathews L, Hong R, Zon G, Brown T, McCormack JJ. Amplification of antibody production by phosphorothioate oligodeoxynucleotides. J. Lab. Clin. Med. 1996;128:329–338. - PubMed
-
- Gursel M, Verthelyi D, Gursel I, Ishii KJ, Klinman DM. Differential and competitive activation of human immune cells by distinct classes of CpG oligodeoxynucleotides. J Leuko Biol. 2002;71:813–820. - PubMed
-
- Halpern MD, Kurlander RJ, Pisetsky DS. Bacterial DNA induces murine interferon-gamma production by stimulation of IL-12 and tumor necrosis factor-alpha. Cell. Immunol. 1996;167:72–78. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

