Utilization of lactose and galactose by Streptococcus mutans: transport, toxicity, and carbon catabolite repression
- PMID: 20190045
- PMCID: PMC2863486
- DOI: 10.1128/JB.01624-09
Utilization of lactose and galactose by Streptococcus mutans: transport, toxicity, and carbon catabolite repression
Abstract
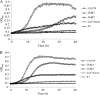
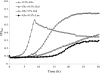
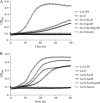
Abundant in milk and other dairy products, lactose is considered to have an important role in oral microbial ecology and can contribute to caries development in both adults and young children. To better understand the metabolism of lactose and galactose by Streptococcus mutans, the major etiological agent of human tooth decay, a genetic analysis of the tagatose-6-phosphate (lac) and Leloir (gal) pathways was performed in strain UA159. Deletion of each gene in the lac operon caused various alterations in expression of a P(lacA)-cat promoter fusion and defects in growth on either lactose (lacA, lacB, lacF, lacE, and lacG), galactose (lacA, lacB, lacD, and lacG) or both sugars (lacA, lacB, and lacG). Failure to grow in the presence of galactose or lactose by certain lac mutants appeared to arise from the accumulation of intermediates of galactose metabolism, particularly galatose-6-phosphate. The glucose- and lactose-PTS permeases, EII(Man) and EII(Lac), respectively, were shown to be the only effective transporters of galactose in S. mutans. Furthermore, disruption of manL, encoding EIIAB(Man), led to increased resistance to glucose-mediated CCR when lactose was used to induce the lac operon, but resulted in reduced lac gene expression in cells growing on galactose. Collectively, the results reveal a remarkably high degree of complexity in the regulation of lactose/galactose catabolism.
Figures
References
-
- Aduse-Opoku, J., L. Tao, J. J. Ferretti, and R. R. Russell. 1991. Biochemical and genetic analysis of Streptococcus mutans α-galactosidase. J. Gen. Microbiol. 137:757-764. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

