In-vitro Release of Rapamycin from a Thermosensitive Polymer for the Inhibition of Vascular Smooth Muscle Cell Proliferation
- PMID: 20190878
- PMCID: PMC2829311
- DOI: 10.4172/jbb.1000002
In-vitro Release of Rapamycin from a Thermosensitive Polymer for the Inhibition of Vascular Smooth Muscle Cell Proliferation
Abstract
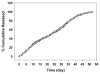
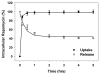
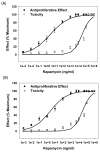
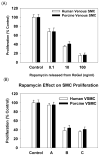
Hemodialysis arteriovenous grafts are often plagued by stenosis at the anastomosis, which is due to the proliferation of vascular smooth muscle cells (SMCs). To prevent the stenosis, we have been developing a strategy for the sustained perivascular delivery of an antiproliferative agent, rapamycin, using an injectable biodegradable polymer, ReGel(®). In this study we examined the in-vitro kinetics of rapamycin released from ReGel and its efficacy for inhibiting the proliferation of human and porcine venous and arterial SMCs. To study the release from ReGel, rapamycin was mixed with ReGel and incubated at 37°C in a release medium. The release medium was periodically sampled and assayed for rapamycin concentration by UV. Cellular uptake and release of rapamycin were examined by incubating SMCs with rapamycin for various durations. Intracellular drug was extracted and measured by HPLC. Antiproliferative effects and cytotoxicity of stock rapamycin and that released from ReGel were examined using cell counting and lactate dehydrogenase (LDH)-release assay, respectively. Rapamycin exhibited a sustained-release pattern from ReGel for 52 days. The kinetics of rapamycin transport through the cell membrane was compatible with a passive diffusion mechanism. Rapamycin released from ReGel exhibited antiproliferative activity similar to the free drug. Our results support the concept of sustained delivery of rapamycin using ReGel as a promising strategy to inhibit SMC proliferation for the prevention of hemodialysis arteriovenous graft stenosis.
Figures
Similar articles
-
Cellular pharmacokinetics and pharmacodynamics of dipyridamole in vascular smooth muscle cells.Biochem Pharmacol. 2006 Oct 16;72(8):956-64. doi: 10.1016/j.bcp.2006.07.027. Epub 2006 Aug 30. Biochem Pharmacol. 2006. PMID: 16939681
-
Inhibition of neointimal hyperplasia in vascular grafts by sustained perivascular delivery of paclitaxel.Kidney Int. 2004 Nov;66(5):2061-9. doi: 10.1111/j.1523-1755.2004.00985.x. Kidney Int. 2004. PMID: 15496180
-
Development of a sustained-release system for perivascular delivery of dipyridamole.J Biomed Mater Res B Appl Biomater. 2006 Apr;77(1):135-43. doi: 10.1002/jbm.b.30412. J Biomed Mater Res B Appl Biomater. 2006. PMID: 16206204
-
Correlation of tissue drug concentrations with in vivo magnetic resonance images of polymer drug depot around arteriovenous graft.J Control Release. 2010 Aug 17;146(1):23-30. doi: 10.1016/j.jconrel.2010.05.005. Epub 2010 May 8. J Control Release. 2010. PMID: 20457189 Free PMC article.
-
Bone morphogenetic protein-7 modulates genes that maintain the vascular smooth muscle cell phenotype in culture.J Bone Joint Surg Am. 2001;83-A Suppl 1(Pt 1):S70-8. J Bone Joint Surg Am. 2001. PMID: 11263669 Review.
Cited by
-
Modelling the Impact of Atherosclerosis on Drug Release and Distribution from Coronary Stents.Ann Biomed Eng. 2016 Feb;44(2):477-87. doi: 10.1007/s10439-015-1456-7. Epub 2015 Sep 18. Ann Biomed Eng. 2016. PMID: 26384667 Free PMC article. Review.
-
Location-dependent coronary artery diffusive and convective mass transport properties of a lipophilic drug surrogate measured using nonlinear microscopy.Pharm Res. 2013 Apr;30(4):1147-60. doi: 10.1007/s11095-012-0950-y. Epub 2012 Dec 7. Pharm Res. 2013. PMID: 23224981 Free PMC article.
-
Periadventitial drug delivery for the prevention of intimal hyperplasia following open surgery.J Control Release. 2016 Jul 10;233:174-80. doi: 10.1016/j.jconrel.2016.05.002. Epub 2016 May 12. J Control Release. 2016. PMID: 27179635 Free PMC article. Review.
-
Concentration-dependent effects of rapamycin on proliferation, migration and apoptosis of endothelial cells in human venous malformation.Exp Ther Med. 2018 Dec;16(6):4595-4601. doi: 10.3892/etm.2018.6782. Epub 2018 Sep 19. Exp Ther Med. 2018. PMID: 30542410 Free PMC article.
-
Rapamycin regulates the expression and activity of Krüppel-like transcription factor 2 in human umbilical vein endothelial cells.PLoS One. 2012;7(8):e43315. doi: 10.1371/journal.pone.0043315. Epub 2012 Aug 24. PLoS One. 2012. PMID: 22937032 Free PMC article.
References
-
- Alexis F, Venkatraman SS, Rath SK, Boey F. In vitro study of release mechanism of paclitaxel and rapamycin from drug-incorporated biodegradable stent matrices. J Control Release. 2004;98:67–74. - PubMed
-
- Charlier A, Leclerc B, Couarraze G. Release of mifepristone from biodegradable matrices: experimental and theoretical evaluations. Int J Pharm. 2000;200:115–20. - PubMed
-
- Gallo R, Padurean A, Jayaraman T, Marx S, Roque M, et al. Inhibition of intimal thickening after balloon angioplasty in porcine coronary arteries by targeting regulators of the cell cycle. Circulation. 1999;99:2164–70. - PubMed
-
- Hille B. Ionic Channels of Excitable Membranes. Sunderland: Sinauer Associates; 1984.
Grants and funding
LinkOut - more resources
Full Text Sources
