Evaluation of formalin inactivated V3526 virus with adjuvant as a next generation vaccine candidate for Venezuelan equine encephalitis virus
- PMID: 20193792
- PMCID: PMC2857980
- DOI: 10.1016/j.vaccine.2010.02.056
Evaluation of formalin inactivated V3526 virus with adjuvant as a next generation vaccine candidate for Venezuelan equine encephalitis virus
Abstract
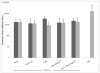
V3526, a genetically modified strain of Venezuelan equine encephalitis virus (VEEV), was formalin inactivated for evaluation as a next generation vaccine candidate for VEEV. In this study, we tested formalin-inactivated V3526 (fV3526) with and without adjuvant for immunogenicity and efficacy in BALB/c mice and results were compared to the existing inactivated VEEV vaccine, C84. Mice were vaccinated intramuscularly (IM) or subcutaneously (SC) with fV3526 formulations and challenged with VEEV IAB Trinidad donkey (VEEV TrD) strain by SC or aerosol exposure. Efficacy following SC or aerosol challenge was not significantly different between the fV3526 formulations or compared to C84 despite C84 being administered in more doses and higher concentration of viral protein per dose. These data support further evaluation of fV3526 formulations as a next generation VEEV vaccine.
Copyright 2010 Elsevier Ltd. All rights reserved.
Figures
Similar articles
-
A multisystem approach for development and evaluation of inactivated vaccines for Venezuelan equine encephalitis virus (VEEV).J Virol Methods. 2010 Feb;163(2):424-32. doi: 10.1016/j.jviromet.2009.11.006. Epub 2009 Nov 10. J Virol Methods. 2010. PMID: 19903494 Free PMC article.
-
Comparison of the immunological responses and efficacy of gamma-irradiated V3526 vaccine formulations against subcutaneous and aerosol challenge with Venezuelan equine encephalitis virus subtype IAB.Vaccine. 2010 Jan 22;28(4):1031-40. doi: 10.1016/j.vaccine.2009.10.126. Epub 2009 Nov 13. Vaccine. 2010. PMID: 19914193 Free PMC article.
-
Venezuelan equine encephalitis virus vaccine candidate (V3526) safety, immunogenicity and efficacy in horses.Vaccine. 2007 Feb 26;25(10):1868-76. doi: 10.1016/j.vaccine.2006.10.030. Epub 2006 Oct 27. Vaccine. 2007. PMID: 17240002 Clinical Trial.
-
Vaccines for Venezuelan equine encephalitis.Vaccine. 2009 Nov 5;27 Suppl 4:D80-5. doi: 10.1016/j.vaccine.2009.07.095. Vaccine. 2009. PMID: 19837294 Free PMC article. Review.
-
A roadmap for developing Venezuelan equine encephalitis virus (VEEV) vaccines: Lessons from the past, strategies for the future.Int J Biol Macromol. 2023 Aug 1;245:125514. doi: 10.1016/j.ijbiomac.2023.125514. Epub 2023 Jun 21. Int J Biol Macromol. 2023. PMID: 37353130 Review.
Cited by
-
Novel vaccine against Venezuelan equine encephalitis combines advantages of DNA immunization and a live attenuated vaccine.Vaccine. 2013 Feb 4;31(7):1019-25. doi: 10.1016/j.vaccine.2012.12.050. Epub 2012 Dec 31. Vaccine. 2013. PMID: 23287629 Free PMC article.
-
Encephalitic Arboviruses: Emergence, Clinical Presentation, and Neuropathogenesis.Neurotherapeutics. 2016 Jul;13(3):514-34. doi: 10.1007/s13311-016-0443-5. Neurotherapeutics. 2016. PMID: 27220616 Free PMC article. Review.
-
Insights into Antibody-Mediated Alphavirus Immunity and Vaccine Development Landscape.Microorganisms. 2021 Apr 22;9(5):899. doi: 10.3390/microorganisms9050899. Microorganisms. 2021. PMID: 33922370 Free PMC article. Review.
-
Therapeutic monoclonal antibody treatment protects nonhuman primates from severe Venezuelan equine encephalitis virus disease after aerosol exposure.PLoS Pathog. 2019 Dec 2;15(12):e1008157. doi: 10.1371/journal.ppat.1008157. eCollection 2019 Dec. PLoS Pathog. 2019. PMID: 31790515 Free PMC article.
-
Current Understanding of the Molecular Basis of Venezuelan Equine Encephalitis Virus Pathogenesis and Vaccine Development.Viruses. 2019 Feb 18;11(2):164. doi: 10.3390/v11020164. Viruses. 2019. PMID: 30781656 Free PMC article. Review.
References
-
- Ehrenkranz NJ, Ventura AK. Venezuelan equine encephalitis virus infection in man. Annu Rev Med. 1974;25:9–14. - PubMed
-
- Hinman AR, McGowan JE, Jr, Henderson BE. Venezuelan equine encephalomyelitis: surveys of human illness during an epizootic in Guatemala and El Salvador. Am J Epidemiol. 1971;93(2):130–6. - PubMed
-
- Smith DG, Mamay HK, Marshall RG, Wagner JC. Venezulean equine encephalomyelitis; laboratory aspects of fourteen human cases following vaccination and attempts to isolate the virus from the vaccine. Am J Hyg. 1956;63(2):150–64. - PubMed
-
- Kortepeter M, Christopher G, Cieslak T, Culpepper R, Darling R. USAMRIID’s Medical Management of Biological Casualities Handbook. 4. Fort Detrick, MD: 2001.
-
- McKinney RW, Berge TO, Sawyer WD, Tigertt WD, Crozier D. Use of an attenuated strain of venezuelan equine encephalomyelitis virus for immunization in man. Am J Trop Med Hyg. 1963;12(4):597–603. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

