Thioredoxin-1 gene therapy enhances angiogenic signaling and reduces ventricular remodeling in infarcted myocardium of diabetic rats
- PMID: 20194885
- PMCID: PMC2852871
- DOI: 10.1161/CIRCULATIONAHA.109.872481
Thioredoxin-1 gene therapy enhances angiogenic signaling and reduces ventricular remodeling in infarcted myocardium of diabetic rats
Abstract
Background: The present study evaluated the reversal of diabetes-mediated impairment of angiogenesis in a myocardial infarction model of type 1 diabetic rats by intramyocardial administration of an adenoviral vector encoding thioredoxin-1 (Ad.Trx1). Various studies have linked diabetes-mediated impairment of angiogenesis to dysfunctional antioxidant systems in which thioredoxin-1 plays a central role.
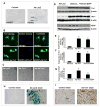
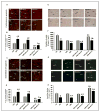
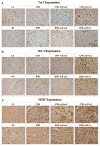
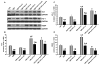
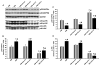
Methods and results: Ad.Trx1 was administered intramyocardially in nondiabetic and diabetic rats immediately after myocardial infarction. Ad.LacZ was similarly administered to the respective control groups. The hearts were excised for molecular and immunohistochemical analysis at predetermined time points. Myocardial function was measured by echocardiography 30 days after the intervention. The Ad.Trx1-administered group exhibited reduced fibrosis, oxidative stress, and cardiomyocyte and endothelial cell apoptosis compared with the diabetic myocardial infarction group, along with increased capillary and arteriolar density. Western blot and immunohistochemical analysis demonstrated myocardial overexpression of thioredoxin-1, heme oxygenase-1, vascular endothelial growth factor, and p38 mitogen-activated protein kinase-beta, as well as decreased phosphorylated JNK and p38 mitogen-activated protein kinase-alpha, in the Ad.Trx1-treated diabetic group. Conversely, we observed a significant reduction in the expression of vascular endothelial growth factor in nondiabetic and diabetic animals treated with tin protoporphyrin (SnPP, a heme oxygenase-1 enzyme inhibitor), even after Ad.Trx1 therapy. Echocardiographic analysis after 4 weeks of myocardial infarction revealed significant improvement in myocardial functional parameters such as ejection fraction, fractional shortening, and E/A ratio in the Ad.Trx1-administered group compared with the diabetic myocardial infarction group.
Conclusions: This study demonstrates for the first time that impairment of angiogenesis and myocardial dysfunction can be regulated by Ad.Trx1 gene therapy in streptozotocin-induced diabetic rats subjected to infarction.
Figures

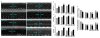
Similar articles
-
Thioredoxin-1 (Trx1) engineered mesenchymal stem cell therapy increased pro-angiogenic factors, reduced fibrosis and improved heart function in the infarcted rat myocardium.Int J Cardiol. 2015 Dec 15;201:517-28. doi: 10.1016/j.ijcard.2015.08.117. Epub 2015 Aug 15. Int J Cardiol. 2015. PMID: 26322599
-
Coadministration of adenoviral vascular endothelial growth factor and angiopoietin-1 enhances vascularization and reduces ventricular remodeling in the infarcted myocardium of type 1 diabetic rats.Diabetes. 2010 Jan;59(1):51-60. doi: 10.2337/db09-0336. Epub 2009 Sep 30. Diabetes. 2010. PMID: 19794062 Free PMC article.
-
Regulation of A-Kinase-Anchoring Protein 12 by Heat Shock Protein A12B to Prevent Ventricular Dysfunction Following Acute Myocardial Infarction in Diabetic Rats.J Cardiovasc Transl Res. 2017 Apr;10(2):209-220. doi: 10.1007/s12265-017-9734-4. Epub 2017 Mar 9. J Cardiovasc Transl Res. 2017. PMID: 28281242
-
Resveratrol enhances neovascularization in the infarcted rat myocardium through the induction of thioredoxin-1, heme oxygenase-1 and vascular endothelial growth factor.J Mol Cell Cardiol. 2005 Nov;39(5):813-22. doi: 10.1016/j.yjmcc.2005.08.003. Epub 2005 Sep 29. J Mol Cell Cardiol. 2005. PMID: 16198371
-
Application of recombinant thioredoxin1 for treatment of heart disease.J Mol Cell Cardiol. 2011 Oct;51(4):570-3. doi: 10.1016/j.yjmcc.2010.09.020. Epub 2010 Oct 16. J Mol Cell Cardiol. 2011. PMID: 20955713 Free PMC article. Review.
Cited by
-
Endothelial dysfunction in diabetes mellitus: possible involvement of endoplasmic reticulum stress?Exp Diabetes Res. 2012;2012:481840. doi: 10.1155/2012/481840. Epub 2012 Feb 28. Exp Diabetes Res. 2012. PMID: 22474423 Free PMC article. Review.
-
Redox balance dynamically regulates vascular growth and remodeling.Semin Cell Dev Biol. 2012 Sep;23(7):745-57. doi: 10.1016/j.semcdb.2012.05.003. Epub 2012 May 24. Semin Cell Dev Biol. 2012. PMID: 22634069 Free PMC article. Review.
-
Diabetic cardiomyopathy and its mechanisms: Role of oxidative stress and damage.J Diabetes Investig. 2014 Nov;5(6):623-34. doi: 10.1111/jdi.12250. Epub 2014 Jul 15. J Diabetes Investig. 2014. PMID: 25422760 Free PMC article.
-
Modulation of signaling mechanisms in the heart by thioredoxin 1.Free Radic Biol Med. 2017 Aug;109:125-131. doi: 10.1016/j.freeradbiomed.2016.12.020. Epub 2016 Dec 16. Free Radic Biol Med. 2017. PMID: 27993729 Free PMC article. Review.
-
The Emerging Role of TXNIP in Ischemic and Cardiovascular Diseases; A Novel Marker and Therapeutic Target.Int J Mol Sci. 2021 Feb 8;22(4):1693. doi: 10.3390/ijms22041693. Int J Mol Sci. 2021. PMID: 33567593 Free PMC article. Review.
References
-
- Barzilay JI, Kronmal RA, Bittner V, Eaker E, Evans C, Foster ED. Coronary artery disease and coronary artery bypass grafting in diabetic patients aged > or = 65 years (report from the Coronary Artery Surgery Study [CASS] Registry) Am J Cardiol. 1994;74:334–339. - PubMed
-
- Martin A, Komada MR, Sane DC. Abnormal angiogenesis in diabetes mellitus. Med Res Rev. 2003;23:117–145. - PubMed
-
- Wiernsperger NF. Oxidative stress as a therapeutic target in diabetes: revisiting the controversy. Diabetes Metab. 2003;29:579–585. - PubMed
-
- Di Filippo C, Cuzzocrea S, Rossi F, Marfella R, D’Amico M. Oxidative stress as the leading cause of acute myocardial infarction in diabetics. Cardiovasc Drug Rev. 2006;24:77–87. - PubMed
-
- Nordberg J, Arner ES. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic Biol Med. 2001;31:1287–1312. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

