Temporal sensitivity of protein kinase a activation in late-phase long term potentiation
- PMID: 20195498
- PMCID: PMC2829045
- DOI: 10.1371/journal.pcbi.1000691
Temporal sensitivity of protein kinase a activation in late-phase long term potentiation
Abstract
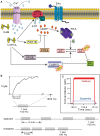
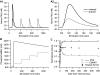
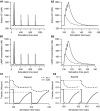
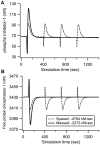
Protein kinases play critical roles in learning and memory and in long term potentiation (LTP), a form of synaptic plasticity. The induction of late-phase LTP (L-LTP) in the CA1 region of the hippocampus requires several kinases, including CaMKII and PKA, which are activated by calcium-dependent signaling processes and other intracellular signaling pathways. The requirement for PKA is limited to L-LTP induced using spaced stimuli, but not massed stimuli. To investigate this temporal sensitivity of PKA, a computational biochemical model of L-LTP induction in CA1 pyramidal neurons was developed. The model describes the interactions of calcium and cAMP signaling pathways and is based on published biochemical measurements of two key synaptic signaling molecules, PKA and CaMKII. The model is stimulated using four 100 Hz tetani separated by 3 sec (massed) or 300 sec (spaced), identical to experimental L-LTP induction protocols. Simulations show that spaced stimulation activates more PKA than massed stimulation, and makes a key experimental prediction, that L-LTP is PKA-dependent for intervals larger than 60 sec. Experimental measurements of L-LTP demonstrate that intervals of 80 sec, but not 40 sec, produce PKA-dependent L-LTP, thereby confirming the model prediction. Examination of CaMKII reveals that its temporal sensitivity is opposite that of PKA, suggesting that PKA is required after spaced stimulation to compensate for a decrease in CaMKII. In addition to explaining the temporal sensitivity of PKA, these simulations suggest that the use of several kinases for memory storage allows each to respond optimally to different temporal patterns.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Colocalization of protein kinase A with adenylyl cyclase enhances protein kinase A activity during induction of long-lasting long-term-potentiation.PLoS Comput Biol. 2011 Jun;7(6):e1002084. doi: 10.1371/journal.pcbi.1002084. Epub 2011 Jun 30. PLoS Comput Biol. 2011. PMID: 21738458 Free PMC article.
-
A model of the roles of essential kinases in the induction and expression of late long-term potentiation.Biophys J. 2006 Apr 15;90(8):2760-75. doi: 10.1529/biophysj.105.072470. Epub 2006 Jan 13. Biophys J. 2006. PMID: 16415049 Free PMC article.
-
Latent Sex Differences in Molecular Signaling That Underlies Excitatory Synaptic Potentiation in the Hippocampus.J Neurosci. 2019 Feb 27;39(9):1552-1565. doi: 10.1523/JNEUROSCI.1897-18.2018. Epub 2018 Dec 21. J Neurosci. 2019. PMID: 30578341 Free PMC article.
-
The molecular basis of CaMKII function in synaptic and behavioural memory.Nat Rev Neurosci. 2002 Mar;3(3):175-90. doi: 10.1038/nrn753. Nat Rev Neurosci. 2002. PMID: 11994750 Review.
-
What does LTP tell us about the roles of CaMKII and PKMζ in memory?Mol Brain. 2018 Dec 28;11(1):77. doi: 10.1186/s13041-018-0420-5. Mol Brain. 2018. PMID: 30593289 Free PMC article. Review.
Cited by
-
Colocalization of protein kinase A with adenylyl cyclase enhances protein kinase A activity during induction of long-lasting long-term-potentiation.PLoS Comput Biol. 2011 Jun;7(6):e1002084. doi: 10.1371/journal.pcbi.1002084. Epub 2011 Jun 30. PLoS Comput Biol. 2011. PMID: 21738458 Free PMC article.
-
A presynaptic role for PKA in synaptic tagging and memory.Neurobiol Learn Mem. 2014 Oct;114:101-112. doi: 10.1016/j.nlm.2014.05.005. Epub 2014 May 29. Neurobiol Learn Mem. 2014. PMID: 24882624 Free PMC article.
-
The right time to learn: mechanisms and optimization of spaced learning.Nat Rev Neurosci. 2016 Feb;17(2):77-88. doi: 10.1038/nrn.2015.18. Nat Rev Neurosci. 2016. PMID: 26806627 Free PMC article. Review.
-
Network motifs exhibiting a differential response to spaced and massed inputs.Learn Mem. 2024 Jul 29;31(7):a054012. doi: 10.1101/lm.054012.124. Print 2024 Jul. Learn Mem. 2024. PMID: 39074905 Free PMC article.
-
FindSim: A Framework for Integrating Neuronal Data and Signaling Models.Front Neuroinform. 2018 Jun 26;12:38. doi: 10.3389/fninf.2018.00038. eCollection 2018. Front Neuroinform. 2018. PMID: 29997492 Free PMC article.
References
-
- Nguyen PV, Woo NH. Regulation of hippocampal synaptic plasticity by cyclic AMP-dependent protein kinases. Prog Neurobiol. 2003;71:401–437. - PubMed
-
- Scharf MT, Woo NH, Lattal KM, Young JZ, Nguyen PV, et al. Protein synthesis is required for the enhancement of long-term potentiation and long-term memory by spaced training. J Neurophysiol. 2002;87:2770–2777. - PubMed
-
- Woo NH, Duffy SN, Abel T, Nguyen PV. Temporal spacing of synaptic stimulation critically modulates the dependence of LTP on cyclic AMP-dependent protein kinase. Hippocampus. 2003;13:293–300. - PubMed
-
- Johnson DA, Akamine P, Radzio-Andzdm E, Madhusudan M, Taylor SS. Dynamics of cAMP-dependent protein kinase. Chem Rev. 2000;101:2243–2270. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

