Blockade of T cell contact-activation of human monocytes by high-density lipoproteins reveals a new pattern of cytokine and inflammatory genes
- PMID: 20195532
- PMCID: PMC2828473
- DOI: 10.1371/journal.pone.0009418
Blockade of T cell contact-activation of human monocytes by high-density lipoproteins reveals a new pattern of cytokine and inflammatory genes
Abstract
Background: Cellular contact with stimulated T cells is a potent inducer of cytokine production in human monocytes and is likely to play a substantial part in chronic/sterile inflammatory diseases. High-density lipoproteins (HDL) specifically inhibit the production of pro-inflammatory cytokines induced by T cell contact.
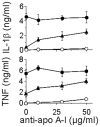
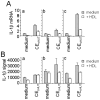
Methodology/principal findings: To further elucidate the pro-inflammatory functions of cellular contact with stimulated T cells and its inhibition by HDL, we carried out multiplex and microarray analyses. Multiplex analysis of monocyte supernatant revealed that 12 out of 27 cytokines were induced upon contact with stimulated T cells, which cytokines included IL-1Ra, G-CSF, GM-CSF, IFNgamma, CCL2, CCL5, TNF, IL-1beta, IL-6, IL-8, CCL3, and CCL4, but only the latter six were inhibited by HDL. Microarray analysis showed that 437 out of 54,675 probe sets were enhanced in monocytes activated by contact with stimulated T cells, 164 probe sets (i.e., 38%) being inhibited by HDL. These results were validated by qPCR. Interestingly, the cytokines induced by T cell contact in monocytes comprised IL-1beta, IL-6 but not IL-12, suggesting that this mechanism might favor Th17 polarization, which emphasizes the relevance of this mechanism to chronic inflammatory diseases and highlights the contrast with acute inflammatory conditions that usually involve lipopolysaccharides (LPS). In addition, the expression of miR-155 and production of prostaglandin E(2)-both involved in inflammatory response-were triggered by T cell contact and inhibited in the presence of HDL.
Conclusions/significance: These results leave no doubt as to the pro-inflammatory nature of T cell contact-activation of human monocytes and the anti-inflammatory functions of HDL.
Conflict of interest statement
Figures
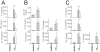


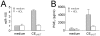
Similar articles
-
HDL interfere with the binding of T cell microparticles to human monocytes to inhibit pro-inflammatory cytokine production.PLoS One. 2010 Jul 29;5(7):e11869. doi: 10.1371/journal.pone.0011869. PLoS One. 2010. PMID: 20686620 Free PMC article.
-
Stimulated T cells generate microparticles, which mimic cellular contact activation of human monocytes: differential regulation of pro- and anti-inflammatory cytokine production by high-density lipoproteins.J Leukoc Biol. 2008 Apr;83(4):921-7. doi: 10.1189/jlb.0807551. Epub 2008 Jan 25. J Leukoc Biol. 2008. PMID: 18223103
-
Differential regulation of cytokine production by PI3Kdelta in human monocytes upon acute and chronic inflammatory conditions.Mol Immunol. 2008 Jul;45(12):3419-27. doi: 10.1016/j.molimm.2008.04.001. Epub 2008 May 9. Mol Immunol. 2008. PMID: 18471882
-
Cytokines, acute-phase proteins, and hormones: IL-1 and TNF-alpha production in contact-mediated activation of monocytes by T lymphocytes.Ann N Y Acad Sci. 2002 Jun;966:464-73. doi: 10.1111/j.1749-6632.2002.tb04248.x. Ann N Y Acad Sci. 2002. PMID: 12114305 Review.
-
Cell contact-mediated signaling of monocytes by stimulated T cells: a major pathway for cytokine induction.Eur Cytokine Netw. 2000 Sep;11(3):346-53. Eur Cytokine Netw. 2000. PMID: 11022117 Review.
Cited by
-
High-density lipoproteins and the immune system.J Lipids. 2013;2013:684903. doi: 10.1155/2013/684903. Epub 2013 Jan 30. J Lipids. 2013. PMID: 23431458 Free PMC article.
-
Longitudinal relationships between rheumatoid factor and cytokine expression by immunostimulated peripheral blood lymphocytes from patients with rheumatoid arthritis: New insights into B-cell activation.Clin Immunol. 2020 Feb;211:108342. doi: 10.1016/j.clim.2020.108342. Epub 2020 Jan 8. Clin Immunol. 2020. PMID: 31926330 Free PMC article.
-
Biomarkers of subclinical atherosclerosis in patients with psoriatic arthritis.Open Access Rheumatol. 2019 Jun 28;11:143-156. doi: 10.2147/OARRR.S206931. eCollection 2019. Open Access Rheumatol. 2019. PMID: 31388317 Free PMC article. Review.
-
HDL Cholesterol as a Marker of Disease Severity and Prognosis in Patients with Pulmonary Arterial Hypertension.Int J Mol Sci. 2019 Jul 18;20(14):3514. doi: 10.3390/ijms20143514. Int J Mol Sci. 2019. PMID: 31323735 Free PMC article. Review.
-
HDL interfere with the binding of T cell microparticles to human monocytes to inhibit pro-inflammatory cytokine production.PLoS One. 2010 Jul 29;5(7):e11869. doi: 10.1371/journal.pone.0011869. PLoS One. 2010. PMID: 20686620 Free PMC article.
References
-
- Vey E, Zhang JH, Dayer JM. IFN-gamma and 1,25(OH)2D3 induce on THP-1 cells distinct patterns of cell surface antigen expression, cytokine production, and responsiveness to contact with activated T cells. J Immunol. 1992;149:2040–2046. - PubMed
-
- Burger D. Cell contact-mediated signaling of monocytes by stimulated T cells: a major pathway for cytokine induction. Eur Cytokine Netw. 2000;11:346–353. - PubMed
-
- Burger D, Dayer JM, Molnarfi N. Cell contact dependence of inflammatory events. In: Smolen JS, Lipsky PE, editors. Contemporary Targeted Therapies in Rheumatology. Abingdon/UK: Taylor & Francis Books Ltd; 2007. pp. 85–103.
-
- Vey E, Dayer JM, Burger D. Direct contact with stimulated T cells induces the expression of IL-1 beta and IL-1 receptor antagonist in human monocytes. Involvement of serine/threonine phosphatases in differential regulation. Cytokine. 1997;9:480–487. - PubMed
-
- Vey E, Burger D, Dayer JM. Expression and cleavage of tumor necrosis factor-alpha and tumor necrosis factor receptors by human monocytic cell lines upon direct contact with stimulated T cells. Eur J Immunol. 1996;26:2404–2409. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

