Microglia activation and anti-inflammatory regulation in Alzheimer's disease
- PMID: 20195797
- PMCID: PMC2892861
- DOI: 10.1007/s12035-010-8106-8
Microglia activation and anti-inflammatory regulation in Alzheimer's disease
Abstract
Inflammatory regulators, including endogenous anti-inflammatory systems, can down-regulate inflammation thus providing negative feedback. Chronic inflammation can result from imbalance between levels of inflammatory mediators and regulators during immune responses. As a consequence, there are heightened inflammatory responses and irreversible tissue damage associated with many age-related chronic diseases. Alzheimer's disease (AD) brain is marked by prominent inflammatory features, in which microglial activation is the driving force for the elaboration of an inflammatory cascade. How the regulation of inflammation loses its effectiveness during AD pathogenesis remains largely unclear. In this article, we will first review current knowledge of microglial activation and its association with AD pathology. We then discuss four examples of anti-inflammatory systems that could play a role in regulating microglial activation: CD200/CD200 receptor, vitamin D receptor, peroxisome proliferator-activated receptors, and soluble receptor for advanced glycation end products. Through this, we hope to illustrate the diverse aspects of inflammatory regulatory systems in brain and neurodegenerative diseases such as AD. We also propose the importance of neuronal defense systems, because they are part of the integral inflammatory and anti-inflammatory systems. Augmenting the anti-inflammatory defenses of neurons can be included in the strategy for restoration of balanced immune responses during aging and neurodegenerative diseases.
Figures

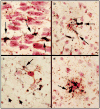
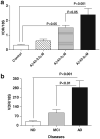
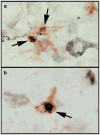

Similar articles
-
Microglia in Alzheimer's disease.Biomed Res Int. 2014;2014:437483. doi: 10.1155/2014/437483. Epub 2014 Aug 14. Biomed Res Int. 2014. PMID: 25197646 Free PMC article. Review.
-
Decreased expression of CD200 and CD200 receptor in Alzheimer's disease: a potential mechanism leading to chronic inflammation.Exp Neurol. 2009 Jan;215(1):5-19. doi: 10.1016/j.expneurol.2008.09.003. Epub 2008 Sep 24. Exp Neurol. 2009. PMID: 18938162 Free PMC article.
-
Modulation of microglial innate immunity in Alzheimer's disease by activation of peroxisome proliferator-activated receptor gamma.Curr Med Chem. 2009;16(6):643-51. doi: 10.2174/092986709787458399. Curr Med Chem. 2009. PMID: 19199928 Free PMC article. Review.
-
The age-related microglial transformation in Alzheimer's disease pathogenesis.Neurobiol Aging. 2020 Aug;92:82-91. doi: 10.1016/j.neurobiolaging.2020.03.024. Epub 2020 Apr 15. Neurobiol Aging. 2020. PMID: 32408056 Review.
-
Preventing activation of receptor for advanced glycation endproducts in Alzheimer's disease.Curr Drug Targets CNS Neurol Disord. 2005 Jun;4(3):249-66. doi: 10.2174/1568007054038210. Curr Drug Targets CNS Neurol Disord. 2005. PMID: 15975028 Review.
Cited by
-
The prenatal challenge with lipopolysaccharide and polyinosinic:polycytidylic acid disrupts CX3CL1-CX3CR1 and CD200-CD200R signalling in the brains of male rat offspring: a link to schizophrenia-like behaviours.J Neuroinflammation. 2020 Aug 23;17(1):247. doi: 10.1186/s12974-020-01923-0. J Neuroinflammation. 2020. PMID: 32829711 Free PMC article.
-
A cytokine mixture of GM-CSF and IL-3 that induces a neuroprotective phenotype of microglia leading to amelioration of (6-OHDA)-induced Parkinsonism of rats.Brain Behav. 2011 Sep;1(1):26-43. doi: 10.1002/brb3.11. Brain Behav. 2011. PMID: 22398979 Free PMC article.
-
MicroRNA-146a switches microglial phenotypes to resist the pathological processes and cognitive degradation of Alzheimer's disease.Theranostics. 2021 Feb 19;11(9):4103-4121. doi: 10.7150/thno.53418. eCollection 2021. Theranostics. 2021. PMID: 33754051 Free PMC article.
-
Deciphering the SUMO code in the kidney.J Cell Mol Med. 2019 Feb;23(2):711-719. doi: 10.1111/jcmm.14021. Epub 2018 Dec 1. J Cell Mol Med. 2019. PMID: 30506859 Free PMC article. Review.
-
Proteomic CNS profile of delayed cognitive impairment in mice exposed to Gulf War agents.Neuromolecular Med. 2011 Dec;13(4):275-88. doi: 10.1007/s12017-011-8160-z. Epub 2011 Oct 11. Neuromolecular Med. 2011. PMID: 21986894
References
-
- Akiyama H, Barger S, Barnum S, Bradt B, Bauer J, Cole GM, Cooper NR, Eikelenboom P, Emmerling M, Fiebich BL, Finch CE, Frautschy S, Griffin WS, Hampel H, Hull M, Landreth G, Lue L, Mrak R, Mackenzie IR, McGeer PL, O'Banion MK, Pachter J, Pasinetti G, Plata-Salaman C, Rogers J, Rydel R, Shen Y, Streit W, Strohmeyer R, Tooyoma I, Van Muiswinkel FL, Veerhuis R, Walker D, Webster S, Wegrzyniak B, Wenk G, Wyss-Coray T. Inflammation and Alzheimer's disease. Neurobiol Aging. 2000;21:383–421. - PMC - PubMed
-
- Walter S, Letiembre M, Liu Y, Heine H, Penke B, Hao W, Bode B, Manietta N, Walter J, Schulz-Schuffer W, Fassbender K. Role of the toll-like receptor 4 in neuroinflammation in Alzheimer's disease. Cell Physiol Biochem. 2007;20:947–956. - PubMed
-
- Hoozemans JJ, Rozemuller JM, van Haastert ES, Veerhuis R, Eikelenboom P. Cyclooxygenase-1 and -2 in the different stages of Alzheimer's disease pathology. Curr Pharm Des. 2008;14:1419–1427. - PubMed
-
- Korotzer AR, Watt J, Cribbs D, Tenner AJ, Burdick D, Glabe C, Cotman CW. Cultured rat microglia express C1q and receptor for C1q: implications for amyloid effects on microglia. Exp Neurol. 1995;134:214–221. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

