Automated electron microscopy for evaluating two-dimensional crystallization of membrane proteins
- PMID: 20197095
- PMCID: PMC2904827
- DOI: 10.1016/j.jsb.2010.02.018
Automated electron microscopy for evaluating two-dimensional crystallization of membrane proteins
Abstract
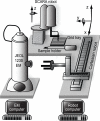
Membrane proteins fulfill many important roles in the cell and represent the target for a large number of therapeutic drugs. Although structure determination of membrane proteins has become a major priority, it has proven to be technically challenging. Electron microscopy of two-dimensional (2D) crystals has the advantage of visualizing membrane proteins in their natural lipidic environment, but has been underutilized in recent structural genomics efforts. To improve the general applicability of electron crystallography, high-throughput methods are needed for screening large numbers of conditions for 2D crystallization, thereby increasing the chances of obtaining well ordered crystals and thus achieving atomic resolution. Previous reports describe devices for growing 2D crystals on a 96-well format. The current report describes a system for automated imaging of these screens with an electron microscope. Samples are inserted with a two-part robot: a SCARA robot for loading samples into the microscope holder, and a Cartesian robot for placing the holder into the electron microscope. A standard JEOL 1230 electron microscope was used, though a new tip was designed for the holder and a toggle switch controlling the airlock was rewired to allow robot control. A computer program for controlling the robots was integrated with the Leginon program, which provides a module for automated imaging of individual samples. The resulting images are uploaded into the Sesame laboratory information management system database where they are associated with other data relevant to the crystallization screen.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures





Similar articles
-
Membrane protein structure determination by electron crystallography.Curr Opin Struct Biol. 2012 Aug;22(4):520-8. doi: 10.1016/j.sbi.2012.04.003. Epub 2012 May 8. Curr Opin Struct Biol. 2012. PMID: 22572457 Free PMC article. Review.
-
Present and future of membrane protein structure determination by electron crystallography.Adv Protein Chem Struct Biol. 2010;81:33-60. doi: 10.1016/B978-0-12-381357-2.00002-5. Adv Protein Chem Struct Biol. 2010. PMID: 21115172 Free PMC article. Review.
-
An automated pipeline to screen membrane protein 2D crystallization.J Struct Funct Genomics. 2010 Jun;11(2):155-66. doi: 10.1007/s10969-010-9088-5. Epub 2010 Mar 27. J Struct Funct Genomics. 2010. PMID: 20349145 Free PMC article.
-
A high-throughput strategy to screen 2D crystallization trials of membrane proteins.J Struct Biol. 2007 Dec;160(3):295-304. doi: 10.1016/j.jsb.2007.09.003. Epub 2007 Sep 14. J Struct Biol. 2007. PMID: 17951070 Free PMC article.
-
High-throughput methods for electron crystallography.Methods Mol Biol. 2013;955:273-96. doi: 10.1007/978-1-62703-176-9_15. Methods Mol Biol. 2013. PMID: 23132066 Free PMC article.
Cited by
-
Inward-facing conformation of the zinc transporter YiiP revealed by cryoelectron microscopy.Proc Natl Acad Sci U S A. 2013 Feb 5;110(6):2140-5. doi: 10.1073/pnas.1215455110. Epub 2013 Jan 22. Proc Natl Acad Sci U S A. 2013. PMID: 23341604 Free PMC article.
-
Specimen preparation for electron diffraction of thin crystals.Micron. 2011 Feb;42(2):132-40. doi: 10.1016/j.micron.2010.05.003. Epub 2010 May 19. Micron. 2011. PMID: 20561794 Free PMC article. Review.
-
Membrane protein structure determination by electron crystallography.Curr Opin Struct Biol. 2012 Aug;22(4):520-8. doi: 10.1016/j.sbi.2012.04.003. Epub 2012 May 8. Curr Opin Struct Biol. 2012. PMID: 22572457 Free PMC article. Review.
-
Present and future of membrane protein structure determination by electron crystallography.Adv Protein Chem Struct Biol. 2010;81:33-60. doi: 10.1016/B978-0-12-381357-2.00002-5. Adv Protein Chem Struct Biol. 2010. PMID: 21115172 Free PMC article. Review.
-
Software tools for automated transmission electron microscopy.Nat Methods. 2019 Jun;16(6):471-477. doi: 10.1038/s41592-019-0396-9. Epub 2019 May 13. Nat Methods. 2019. PMID: 31086343 Free PMC article. Review.
References
-
- Sugahara M, Asada Y, Shimizu K, Yamamoto H, Lokanath NK, Mizutani H, Bagautdinov B, Matsuura Y, Taketa M, Kageyama Y, Ono N, Morikawa Y, Tanaka Y, Shimada H, Nakamoto T, Yamamoto M, Kunishima N. High-throughput crystallization-to-structure pipeline at RIKEN SPring-8 Center. J Struct Funct Genomics. 2008;9:21–8. - PubMed
-
- Wiener MC. A pedestrian guide to membrane protein crystallization. Methods. 2004;34:364–72. - PubMed
-
- Amos LA, Henderson R, Unwin PNT. Three-dimensional structure determination by electron microscopy of two-dimensional crystals. Prog. Biophys. Molec. Biol. 1982;39:183–231. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

