Inhibition of bradykinin receptor B1 protects mice from focal brain injury by reducing blood-brain barrier leakage and inflammation
- PMID: 20197781
- PMCID: PMC2949241
- DOI: 10.1038/jcbfm.2010.28
Inhibition of bradykinin receptor B1 protects mice from focal brain injury by reducing blood-brain barrier leakage and inflammation
Abstract
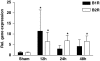
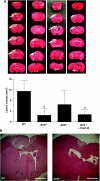
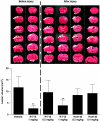
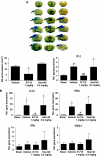
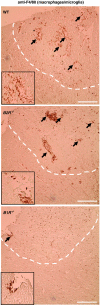
Kinins are proinflammatory and vasoactive peptides that are released during tissue damage and may contribute to neuronal degeneration, inflammation, and edema formation after brain injury by acting on discrete bradykinin receptors, B1R and B2R. We studied the expression of B1R and B2R and the effect of their inhibition on lesion size, blood-brain barrier (BBB) disruption, and inflammatory processes after a focal cryolesion of the right parietal cortex in mice. B1R and B2R gene transcripts were significantly induced in the lesioned hemispheres of wild-type mice (P<0.05). The volume of the cortical lesions and neuronal damage at 24 h after injury in B1R(-/-) mice were significantly smaller than in wild-type controls (2.5+/-2.6 versus 11.5+/-3.9 mm(3), P<0.001). Treatment with the B1R antagonist R-715 1 h after lesion induction likewise reduced lesion volume in wild-type mice (2.6+/-1.4 versus 12.2+/-6.1 mm(3), P<0.001). This was accompanied by a remarkable reduction of BBB disruption and tissue inflammation. In contrast, genetic deletion or pharmacological inhibition of B2R had no significant impact on lesion formation or the development of brain edema. We conclude that B1R inhibition may offer a novel therapeutic strategy after acute brain injuries.
Figures
References
-
- Austinat M, Braeuninger S, Pesquero JB, Brede M, Bader M, Stoll G, Renne T, Kleinschnitz C. Blockade of bradykinin receptor B1 but not bradykinin receptor B2 provides protection from cerebral infarction and brain edema. Stroke. 2009;40:285–293. - PubMed
-
- Camargo AC, Shapanka R, Greene LJ. Preparation, assay, and partial characterization of a neutral endopeptidase from rabbit brain. Biochemistry. 1973;129:1838–1844. - PubMed
-
- Gobeil F, Neugebauer W, Filteau C, Jukic D, Allogho SN, Pheng LH, Nguyen-Le XK, Blouin D, Regoli D. Structure-activity studies of B1 receptor-related peptides. Antagonists. Hypertension. 1996;28:833–839. - PubMed
-
- Gorlach C, Hortobagyi T, Hortobagyi S, Benyo Z, Relton J, Whalley ET, Wahl M. Bradykinin B2, but not B1, receptor antagonism has a neuroprotective effect after brain injury. J Neurotrauma. 2001;18:833–838. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

