Activation of PKC isoform beta(I) at the blood-brain barrier rapidly decreases P-glycoprotein activity and enhances drug delivery to the brain
- PMID: 20197783
- PMCID: PMC2949219
- DOI: 10.1038/jcbfm.2010.21
Activation of PKC isoform beta(I) at the blood-brain barrier rapidly decreases P-glycoprotein activity and enhances drug delivery to the brain
Abstract
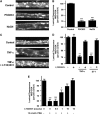
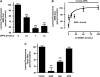
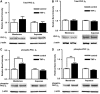
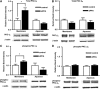
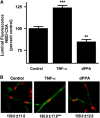
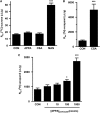
P-glycoprotein is an ATP (adenosine triphosphate)-driven drug efflux transporter that is highly expressed at the blood-brain barrier (BBB) and is a major obstacle to the pharmacotherapy of central nervous system diseases, including brain tumors, neuro-AIDS, and epilepsy. Previous studies have shown that P-glycoprotein transport activity in rat brain capillaries is rapidly reduced by the proinflammatory cytokine, tumor necrosis factor-alpha (TNF-alpha) acting through protein kinase C (PKC)-dependent signaling. In this study, we used isolated rat brain capillaries to show that the TNF-alpha-induced reduction of P-glycoprotein activity was prevented by a PKCbeta(I/II) inhibitor, LY333531, and mimicked by a PKCbeta(I/II) activator, 12-deoxyphorbol-13-phenylacetate-20-acetate (dPPA). Western blotting of brain capillary extracts with phospho-specific antibodies showed that dPPA activated PKCbeta(I), but not PKCbeta(II). Moreover, in intact rats, intracarotid infusion of dPPA potently increased brain accumulation of the P-glycoprotein substrate, [(3)H]-verapamil without compromising tight junction integrity. Thus, PKCbeta(I) activation selectively reduced P-glycoprotein activity both in vitro and in vivo. Targeting PKCbeta(I) at the BBB may prove to be an effective strategy for enhancing the delivery of small molecule therapeutics to the brain.
Figures
Similar articles
-
Targeting blood-brain barrier sphingolipid signaling reduces basal P-glycoprotein activity and improves drug delivery to the brain.Proc Natl Acad Sci U S A. 2012 Sep 25;109(39):15930-5. doi: 10.1073/pnas.1203534109. Epub 2012 Sep 4. Proc Natl Acad Sci U S A. 2012. PMID: 22949658 Free PMC article.
-
Activating PKC-β1 at the blood-brain barrier reverses induction of P-glycoprotein activity by dioxin and restores drug delivery to the CNS.J Cereb Blood Flow Metab. 2011 Jun;31(6):1371-5. doi: 10.1038/jcbfm.2011.44. Epub 2011 Apr 13. J Cereb Blood Flow Metab. 2011. PMID: 21487415 Free PMC article.
-
Rapid, reversible modulation of blood-brain barrier P-glycoprotein transport activity by vascular endothelial growth factor.J Neurosci. 2010 Jan 27;30(4):1417-25. doi: 10.1523/JNEUROSCI.5103-09.2010. J Neurosci. 2010. PMID: 20107068 Free PMC article.
-
Modulation of P-glycoprotein at the blood-brain barrier: opportunities to improve central nervous system pharmacotherapy.Pharmacol Rev. 2008 Jun;60(2):196-209. doi: 10.1124/pr.107.07109. Epub 2008 Jun 17. Pharmacol Rev. 2008. PMID: 18560012 Free PMC article. Review.
-
PET Studies on P-glycoprotein function in the blood-brain barrier: how it affects uptake and binding of drugs within the CNS.Curr Pharm Des. 2004;10(13):1493-503. doi: 10.2174/1381612043384736. Curr Pharm Des. 2004. PMID: 15134571 Review.
Cited by
-
Distinct roles of ezrin, radixin and moesin in maintaining the plasma membrane localizations and functions of human blood-brain barrier transporters.J Cereb Blood Flow Metab. 2020 Jul;40(7):1533-1545. doi: 10.1177/0271678X19868880. Epub 2019 Aug 14. J Cereb Blood Flow Metab. 2020. PMID: 31409174 Free PMC article.
-
Protein kinase Cβ as a therapeutic target stabilizing blood-brain barrier disruption in experimental autoimmune encephalomyelitis.Proc Natl Acad Sci U S A. 2013 Sep 3;110(36):14735-40. doi: 10.1073/pnas.1302569110. Epub 2013 Aug 19. Proc Natl Acad Sci U S A. 2013. PMID: 23959874 Free PMC article.
-
Mrp1 is essential for sphingolipid signaling to p-glycoprotein in mouse blood-brain and blood-spinal cord barriers.J Cereb Blood Flow Metab. 2013 Mar;33(3):381-8. doi: 10.1038/jcbfm.2012.174. Epub 2012 Nov 21. J Cereb Blood Flow Metab. 2013. PMID: 23168528 Free PMC article.
-
Aryl hydrocarbon receptor-mediated up-regulation of ATP-driven xenobiotic efflux transporters at the blood-brain barrier.FASEB J. 2011 Feb;25(2):644-52. doi: 10.1096/fj.10-169227. Epub 2010 Nov 3. FASEB J. 2011. PMID: 21048045 Free PMC article.
-
Effects of hepatic ischemia-reperfusion injury on the P-glycoprotein activity at the liver canalicular membrane and blood-brain barrier determined by in vivo administration of rhodamine 123 in rats.Pharm Res. 2014 Apr;31(4):861-73. doi: 10.1007/s11095-013-1208-z. Epub 2013 Sep 25. Pharm Res. 2014. PMID: 24065596
References
-
- Bauer B, Hartz AM, Lucking JR, Yang X, Pollack GM, Miller DS. Coordinated nuclear receptor regulation of the efflux transporter, Mrp2, and the phase-II metabolizing enzyme, GSTpi, at the blood-brain barrier. J Cereb Blood Flow Metab. 2008a;28:1222–1234. - PubMed
-
- Bauer B, Hartz AM, Miller DS. Tumor necrosis factor alpha and endothelin-1 increase P-glycoprotein expression and transport activity at the blood-brain barrier. Mol Pharmacol. 2007;71:667–675. - PubMed
-
- Bauer B, Hartz AM, Pekcec A, Toellner K, Miller DS, Potschka H. Seizure-induced up-regulation of P-glycoprotein at the blood-brain barrier through glutamate and cyclooxygenase-2 signaling. Mol Pharmacol. 2008b;73:1444–1453. - PubMed
-
- Broughman JR, Sun L, Umar S, Scott J, Sellin JH, Morris AP. Chronic PKC-beta activation in HT-29 Cl.19a colonocytes prevents cAMP-mediated ion secretion by inhibiting apical membrane current generation. Am J Physiol. 2006;291:G318–G330. - PubMed
-
- Brown RC, Egleton RD, Davis TP. Mannitol opening of the blood-brain barrier: regional variation in the permeability of sucrose, but not 86Rb+ or albumin. Brain Res. 2004;1014:221–227. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

