Parasite virulence when the infection reduces the host immune response
- PMID: 20200031
- PMCID: PMC2871884
- DOI: 10.1098/rspb.2010.0138
Parasite virulence when the infection reduces the host immune response
Abstract
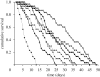
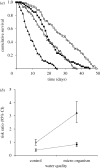
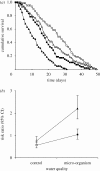
Parasite infections often induce a reduction in host immune response either because of a direct manipulation of the immune system by the parasite or because of energy depletion. Although infection-induced immunodepression can favour the establishment of the parasite within the host, a too severe immunodepression may increase the risk of infection with opportunistic pathogens, stopping the period over which the parasite can be transmitted to other hosts. Here, we explore how the risk of contracting opportunistic diseases affects the survival of the amphipod Gammarus pulex infected by the acanthocephalan Pomphorhynchus laevis. Previous work with this system has shown that upon infection, G. pulex has a substantially reduced immune response. Non-infected and P. laevis-infected hosts were maintained either in control or in micro-organism-enriched water, so as to vary the risk of encountering opportunistic pathogens. As predicted, we found that host mortality was exacerbated when infected gammarids were maintained in micro-organism-enriched water compared with clean, control water; whereas for non-infected gammarids, living in micro-organism-enriched water only moderately increased the risk of mortality. These results show that the virulence of parasites that reduce the host immune response is an environmentally sensitive trait that depends on the concomitant risk for the host of contracting opportunistic diseases. This extra source of host mortality probably represents a cost for P. laevis, and we tentatively predict that the optimal level of parasite exploitation should depend on environmental conditions.
Figures
Similar articles
-
Density-dependent effects on parasite growth and parasite-induced host immunodepression in the larval helminth Pomphorhynchus laevis.Parasitology. 2011 Feb;138(2):257-65. doi: 10.1017/S0031182010001083. Epub 2010 Aug 10. Parasitology. 2011. PMID: 20696096
-
Variation and covariation in infectivity, virulence and immunodepression in the host-parasite association Gammarus pulex-Pomphorhynchus laevis.Proc Biol Sci. 2009 Dec 7;276(1676):4229-36. doi: 10.1098/rspb.2009.1299. Epub 2009 Sep 2. Proc Biol Sci. 2009. PMID: 19726474 Free PMC article.
-
Parasite-induced alteration of plastic response to predation threat: increased refuge use but lower food intake in Gammarus pulex infected with the acanothocephalan Pomphorhynchus laevis.Int J Parasitol. 2014 Mar;44(3-4):211-6. doi: 10.1016/j.ijpara.2013.11.001. Epub 2013 Dec 1. Int J Parasitol. 2014. PMID: 24291320
-
Magnitude and direction of parasite-induced phenotypic alterations: a meta-analysis in acanthocephalans.Biol Rev Camb Philos Soc. 2020 Oct;95(5):1233-1251. doi: 10.1111/brv.12606. Epub 2020 Apr 27. Biol Rev Camb Philos Soc. 2020. PMID: 32342653 Review.
-
Amphipod parasites may bias results of ecotoxicological research.Dis Aquat Organ. 2019 Oct 2;136(1):123-134. doi: 10.3354/dao03355. Dis Aquat Organ. 2019. PMID: 31575839 Review.
Cited by
-
Parasite resource manipulation drives bimodal variation in infection duration.Proc Biol Sci. 2019 May 15;286(1902):20190456. doi: 10.1098/rspb.2019.0456. Proc Biol Sci. 2019. PMID: 31064304 Free PMC article.
-
Avian haemosporidian persistence and co-infection in great tits at the individual level.Malar J. 2013 Jan 30;12:40. doi: 10.1186/1475-2875-12-40. Malar J. 2013. PMID: 23360530 Free PMC article.
-
Immune evasion, immunopathology and the regulation of the immune system.Pathogens. 2013 Feb 13;2(1):71-91. doi: 10.3390/pathogens2010071. Pathogens. 2013. PMID: 25436882 Free PMC article. Review.
-
Immune gene expression in the mosquito vector Culex quinquefasciatus during an avian malaria infection.Mol Ecol. 2023 Feb;32(4):904-919. doi: 10.1111/mec.16799. Epub 2022 Dec 14. Mol Ecol. 2023. PMID: 36448733 Free PMC article.
-
A large-scale epidemiological investigation on trematode infections in small ruminants in Bangladesh.Vet Med Sci. 2022 May;8(3):1219-1228. doi: 10.1002/vms3.748. Epub 2022 Feb 3. Vet Med Sci. 2022. PMID: 35113500 Free PMC article.
References
-
- Benesh D. P., Valtonen E. T.2007Effects of Acanthocephalus lucii (Acanthocephala) on intermediate host survival and growth: implications for exploitation strategies. J. Parasitol. 93, 735–741 (doi:10.1645/GE-1093R.1) - DOI - PubMed
-
- Bethel W. M., Holmes J. C.1974Correlation of development of altered evasive behaviour in Gammrus lacustris (Amphipoda) harboring cystacanths of Polymorphus paradoxus (Acanthocephala) with the infectivity to the definitive host. J. Parasitol. 60, 272–274 (doi:10.2307/3278463) - DOI - PubMed
-
- Bollache L., Rigaud T., Cézilly F.2002Effects of two acanthocephalan parasites on the fecundity and pairing status of female Gammarus pulex (Crustacea: Amphipoda). J. Invertebr. Pathol. 79, 102–110 (doi:10.1016/S0022-2011(02)00027-7) - DOI - PubMed
-
- Cerenius L., Söderhäll K.2004The prophenoloxidase-activating system in invertebrates. Immunol. Rev. 198, 116–126 (doi:10.1111/j.0105-2896.2004.00116.x) - DOI - PubMed
-
- Cornet S., Franceschi N., Bauer A., Rigaud T., Moret Y.2009aImmune depression induced by acanthocephalan parasites in their intermediate crustacean host: consequences for the risk of super-infection and links with host behavioural manipulation. Int. J. Parasitol. 39, 221–229 (doi:10.1016/j.ijpara.2008.06.007) - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

