Electrostatic surface modifications to improve gene delivery
- PMID: 20201712
- PMCID: PMC2911010
- DOI: 10.1517/17425241003603653
Electrostatic surface modifications to improve gene delivery
Abstract
Importance of the field: Gene therapy has the potential to treat a wide variety of diseases, including genetic diseases and cancer.
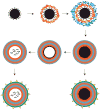
Areas covered in this review: This review introduces biomaterials used for gene delivery and then focuses on the use of electrostatic surface modifications to improve gene delivery materials. These modifications have been used to stabilize therapeutics in vivo, add cell-specific targeting ligands, and promote controlled release. Coatings of nanoparticles and microparticles as well as non-particulate surface coatings are covered in this review. Electrostatic principles are crucial for the development of multilayer delivery structures fabricated by the layer-by-layer method.
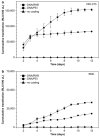
What the reader will gain: The reader will gain knowledge about the composition of biomaterials used for surface modifications and how these coatings and multilayers can be utilized to improve spatial control and efficiency of delivery. Examples are shown for the delivery of nucleic acids, including DNA and siRNA, to in vitro and in vivo systems.
Take home message: The versatile and powerful approach of electrostatic coatings and multilayers will lead to the development of enhanced gene therapies.
Figures




Similar articles
-
Substrate-mediated nucleic acid delivery from self-assembled monolayers.Trends Biotechnol. 2011 Mar;29(3):119-26. doi: 10.1016/j.tibtech.2010.11.005. Epub 2011 Jan 3. Trends Biotechnol. 2011. PMID: 21208672 Review.
-
Biomaterial-based delivery of nucleic acids for tissue regeneration.Adv Drug Deliv Rev. 2021 Sep;176:113885. doi: 10.1016/j.addr.2021.113885. Epub 2021 Jul 27. Adv Drug Deliv Rev. 2021. PMID: 34324886 Review.
-
Development and functionality analysis of lipoplex-loaded polysaccharide-based surface coatings for local nucleic acid delivery.Int J Pharm. 2025 Mar 30;673:125330. doi: 10.1016/j.ijpharm.2025.125330. Epub 2025 Feb 14. Int J Pharm. 2025. PMID: 39956410
-
Non-viral nucleic acid containing nanoparticles as cancer therapeutics.Expert Opin Drug Deliv. 2016 Oct;13(10):1475-87. doi: 10.1080/17425247.2016.1190707. Epub 2016 Jun 6. Expert Opin Drug Deliv. 2016. PMID: 27248202 Free PMC article. Review.
-
Recent developments in nucleic acid delivery with polyethylenimines.Adv Genet. 2014;88:263-88. doi: 10.1016/B978-0-12-800148-6.00009-2. Adv Genet. 2014. PMID: 25409609 Review.
Cited by
-
Uptake and transfection with polymeric nanoparticles are dependent on polymer end-group structure, but largely independent of nanoparticle physical and chemical properties.Mol Pharm. 2012 Nov 5;9(11):3375-83. doi: 10.1021/mp3004176. Epub 2012 Sep 27. Mol Pharm. 2012. PMID: 22970908 Free PMC article.
-
Serum-Resistant Ternary DNA Polyplexes for Suicide Gene Therapy of Uterine Leiomyoma.Int J Mol Sci. 2023 Dec 19;25(1):34. doi: 10.3390/ijms25010034. Int J Mol Sci. 2023. PMID: 38203202 Free PMC article.
-
2011 Rita Schaffer lecture: nanoparticles for intracellular nucleic acid delivery.Ann Biomed Eng. 2012 Jul;40(7):1408-18. doi: 10.1007/s10439-012-0550-3. Epub 2012 Mar 27. Ann Biomed Eng. 2012. PMID: 22451256 Free PMC article.
-
Hitting Undruggable Targets: Viewing Stabilized Peptide Development through the Lens of Quantitative Systems Pharmacology.Trends Biochem Sci. 2019 Mar;44(3):241-257. doi: 10.1016/j.tibs.2018.11.008. Epub 2018 Dec 15. Trends Biochem Sci. 2019. PMID: 30563724 Free PMC article. Review.
-
Cooperativity Principles in Self-Assembled Nanomedicine.Chem Rev. 2018 Jun 13;118(11):5359-5391. doi: 10.1021/acs.chemrev.8b00195. Epub 2018 Apr 25. Chem Rev. 2018. PMID: 29693377 Free PMC article. Review.
References
-
- Langer R, Tirrell DA. Designing materials for biology and medicine. Nature. 2004;428(6982):487–92. - PubMed
-
- Hubbell J. Bioactive biomaterials. Curr Opin Biotechnol. 1999;10(2):123–9. - PubMed
-
- Godbey WT, Barry MA, Saggau P, et al. Poly(ethylenimine)-mediated transfection: a new paradigm for gene delivery. J Biomed Mater Res. 2000;51(3):321–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
