Effect of indomethacin on bile acid-phospholipid interactions: implication for small intestinal injury induced by nonsteroidal anti-inflammatory drugs
- PMID: 20203063
- PMCID: PMC2867422
- DOI: 10.1152/ajpgi.00387.2009
Effect of indomethacin on bile acid-phospholipid interactions: implication for small intestinal injury induced by nonsteroidal anti-inflammatory drugs
Abstract
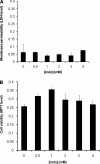
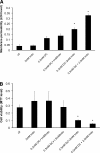
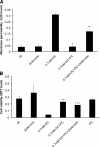
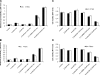
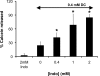
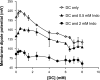
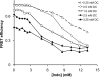
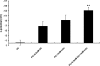
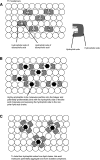
The injurious effect of nonsteroidal anti-inflammatory drugs (NSAIDs) in the small intestine was not appreciated until the widespread use of capsule endoscopy. Animal studies found that NSAID-induced small intestinal injury depends on the ability of these drugs to be secreted into the bile. Because the individual toxicity of amphiphilic bile acids and NSAIDs directly correlates with their interactions with phospholipid membranes, we propose that the presence of both NSAIDs and bile acids alters their individual physicochemical properties and enhances the disruptive effect on cell membranes and overall cytotoxicity. We utilized in vitro gastric AGS and intestinal IEC-6 cells and found that combinations of bile acid, deoxycholic acid (DC), taurodeoxycholic acid, glycodeoxycholic acid, and the NSAID indomethacin (Indo) significantly increased cell plasma membrane permeability and became more cytotoxic than these agents alone. We confirmed this finding by measuring liposome permeability and intramembrane packing in synthetic model membranes exposed to DC, Indo, or combinations of both agents. By measuring physicochemical parameters, such as fluorescence resonance energy transfer and membrane surface charge, we found that Indo associated with phosphatidylcholine and promoted the molecular aggregation of DC and potential formation of larger and isolated bile acid complexes within either biomembranes or bile acid-lipid mixed micelles, which leads to membrane disruption. In this study, we demonstrated increased cytotoxicity of combinations of bile acid and NSAID and provided a molecular mechanism for the observed toxicity. This mechanism potentially contributes to the NSAID-induced injury in the small bowel.
Figures
References
-
- Bjarnason I, Hayllar J, MacPherson AJ, Russell AS. Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology 104: 1832–1847, 1993 - PubMed
-
- Brune K, Nurnberg B, Szelenyi I. The Enterohepatic Circulation of Some Anti-Inflammatory Drugs May Cause Intestinal Ulcerations Boston, MA: MTP Press, 1985
-
- Carey MC, Montet JC, Phillips MC, Armstrong MJ, Mazer NA. Thermodynamic and molecular basis for dissimilar cholesterol-solubilizing capacities by micellar solutions of bile salts: cases of sodium chenodeoxycholate and sodium ursodeoxycholate and their glycine and taurine conjugates. Biochemistry 20: 3637–3648, 1981 - PubMed
-
- Carey MC, Small DM. Micellar properties of dihydroxy and trihydroxy bile salts: effects of counterion and temperature. J Colloid Interface Sci 31: 382–396, 1969 - PubMed
-
- Clarke RJ. Effect of lipid structure on the dipole potential of phosphatidylcholine bilayers. Biochim Biophys Acta 1327: 269–278, 1997 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

