Striatal muscarinic receptors promote activity dependence of dopamine transmission via distinct receptor subtypes on cholinergic interneurons in ventral versus dorsal striatum
- PMID: 20203199
- PMCID: PMC2866006
- DOI: 10.1523/JNEUROSCI.5620-09.2010
Striatal muscarinic receptors promote activity dependence of dopamine transmission via distinct receptor subtypes on cholinergic interneurons in ventral versus dorsal striatum
Abstract
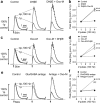
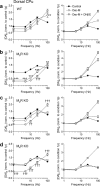
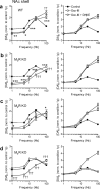
Striatal dopamine (DA) and acetylcholine (ACh) regulate motivated behaviors and striatal plasticity. Interactions between these neurotransmitters may be important, through synchronous changes in parent neuron activities and reciprocal presynaptic regulation of release. How DA signaling is regulated by striatal muscarinic receptors (mAChRs) is unresolved; contradictory reports indicate suppression or facilitation, implicating several mAChR subtypes on various neurons. We investigated whether mAChR regulation of DA signaling varies with presynaptic activity and identified the mAChRs responsible in sensorimotor- versus limbic-associated striatum. We detected DA in real time at carbon fiber microelectrodes in mouse striatal slices. Broad-spectrum mAChR agonists [oxotremorine-M, APET (arecaidine propargyl ester tosylate)] decreased DA release evoked by low-frequency stimuli (1-10 Hz, four pulses) but increased the sensitivity of DA release to presynaptic activity, even enhancing release by high frequencies (e.g., >25 Hz for four pulses). These bidirectional effects depended on ACh input to striatal nicotinic receptors (nAChRs) on DA axons but not GABA or glutamate input. In caudate-putamen (CPu), knock-out of M(2)- or M(4)-mAChRs (not M(5)) prevented mAChR control of DA, indicating that M(2)- and M(4)-mAChRs are required. In nucleus accumbens (NAc) core or shell, mAChR function was prevented in M(4)-knock-outs, but not M(2)- or M(5)-knock-outs. These data indicate that striatal mAChRs, by inhibiting ACh release from cholinergic interneurons and thus modifying nAChR activity, offer variable control of DA release probability that promotes how DA release reflects activation of dopaminergic axons. Furthermore, different coupling of striatal M(2)/M(4)-mAChRs to the control of DA release in CPu versus NAc suggests targets to influence DA/ACh function differentially between striatal domains.
Figures




Similar articles
-
Regulation of glutamate release from primary afferents and interneurons in the spinal cord by muscarinic receptor subtypes.J Neurophysiol. 2007 Jan;97(1):102-9. doi: 10.1152/jn.00586.2006. Epub 2006 Oct 18. J Neurophysiol. 2007. PMID: 17050831
-
Multiple muscarinic acetylcholine receptor subtypes modulate striatal dopamine release, as studied with M1-M5 muscarinic receptor knock-out mice.J Neurosci. 2002 Aug 1;22(15):6347-52. doi: 10.1523/JNEUROSCI.22-15-06347.2002. J Neurosci. 2002. PMID: 12151512 Free PMC article.
-
Nitric oxide donors enhance the frequency dependence of dopamine release in nucleus accumbens.Neuropsychopharmacology. 2011 Aug;36(9):1811-22. doi: 10.1038/npp.2011.62. Epub 2011 Apr 20. Neuropsychopharmacology. 2011. PMID: 21508928 Free PMC article.
-
Roles of the M4 acetylcholine receptor in the basal ganglia and the treatment of movement disorders.Mov Disord. 2019 Aug;34(8):1089-1099. doi: 10.1002/mds.27740. Epub 2019 Jun 18. Mov Disord. 2019. PMID: 31211471 Free PMC article. Review.
-
Cholinergic interneuron characteristics and nicotinic properties in the striatum.J Neurobiol. 2002 Dec;53(4):590-605. doi: 10.1002/neu.10150. J Neurobiol. 2002. PMID: 12436423 Review.
Cited by
-
Opposing regulation of dopaminergic activity and exploratory motor behavior by forebrain and brainstem cholinergic circuits.Nat Commun. 2012;3:1172. doi: 10.1038/ncomms2144. Nat Commun. 2012. PMID: 23132022 Free PMC article.
-
The neurobiological basis for novel experimental therapeutics in dystonia.Neurobiol Dis. 2019 Oct;130:104526. doi: 10.1016/j.nbd.2019.104526. Epub 2019 Jul 4. Neurobiol Dis. 2019. PMID: 31279827 Free PMC article. Review.
-
Complex Control of Striatal Neurotransmission by Nicotinic Acetylcholine Receptors via Excitatory Inputs onto Medium Spiny Neurons.J Neurosci. 2018 Jul 18;38(29):6597-6607. doi: 10.1523/JNEUROSCI.0071-18.2018. Epub 2018 Jun 25. J Neurosci. 2018. PMID: 29941445 Free PMC article.
-
Targeting the Actions of Muscarinic Receptors on Dopamine Systems: New Strategies for Treating Neuropsychiatric Disorders.Annu Rev Pharmacol Toxicol. 2024 Jan 23;64:277-289. doi: 10.1146/annurev-pharmtox-051921-023858. Epub 2023 Aug 9. Annu Rev Pharmacol Toxicol. 2024. PMID: 37552895 Free PMC article. Review.
-
Contribution of both M1 and M4 receptors to muscarinic agonist-mediated attenuation of the cocaine discriminative stimulus in mice.Psychopharmacology (Berl). 2012 Apr;220(4):673-85. doi: 10.1007/s00213-011-2516-9. Epub 2011 Oct 1. Psychopharmacology (Berl). 2012. PMID: 21964721 Free PMC article.
References
-
- Abeliovich A, Schmitz Y, Fariñas I, Choi-Lundberg D, Ho WH, Castillo PE, Shinsky N, Verdugo JM, Armanini M, Ryan A, Hynes M, Phillips H, Sulzer D, Rosenthal A. Mice lacking alpha-synuclein display functional deficits in the nigrostriatal dopamine system. Neuron. 2000;25:239–252. - PubMed
-
- Alcantara AA, Mrzljak L, Jakab RL, Levey AI, Hersch SM, Goldman-Rakic PS. Muscarinic m1 and m2 receptor proteins in local circuit and projection neurons of the primate striatum: anatomical evidence for cholinergic modulation of glutamatergic prefronto-striatal pathways. J Comp Neurol. 2001;434:445–460. - PubMed
-
- Apicella P. Tonically active neurons in the primate striatum and their role in the processing of information about motivationally relevant events. Eur J Neurosci. 2002;16:2017–2026. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
