Mechanisms of tumor necrosis factor-alpha-induced interleukin-6 synthesis in glioma cells
- PMID: 20205746
- PMCID: PMC2846903
- DOI: 10.1186/1742-2094-7-16
Mechanisms of tumor necrosis factor-alpha-induced interleukin-6 synthesis in glioma cells
Abstract
Background: Interleukin (IL)-6 plays a pivotal role in a variety of CNS functions such as the induction and modulation of reactive astrogliosis, pathological inflammatory responses and neuroprotection. Tumor necrosis factor (TNF)-alpha induces IL-6 release from rat C6 glioma cells through the inhibitory kappa B (IkappaB)-nuclear factor kappa B (NFkappaB) pathway, p38 mitogen-activated protein (MAP) kinase and stress-activated protein kinase (SAPK)/c-Jun N-terminal kinase (JNK). The present study investigated the mechanism of TNF-alpha-induced IL-6 release in more detail than has previously been reported.
Methods: Cultured C6 cells were stimulated by TNF-alpha. IL-6 release from the cells was measured by an enzyme-linked immunosorbent assay, and the phosphorylation of IkappaB, NFkappaB, the MAP kinase superfamily, and signal transducer and activator of transcription (STAT)3 was analyzed by Western blotting. Levels of IL-6 mRNA in cells were evaluated by real-time reverse transcription-polymerase chain reaction.
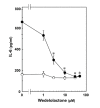
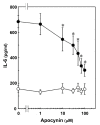
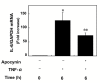
Results: TNF-alpha significantly induced phosphorylation of NFkappaB at Ser 536 and Ser 468, but not at Ser 529 or Ser 276. Wedelolactone, an inhibitor of IkappaB kinase, suppressed both TNF-alpha-induced IkappaB phosphorylation and NFkappaB phosphorylation at Ser 536 and Ser 468. TNF-alpha-stimulated increases in IL-6 levels were suppressed by wedelolactone. TNF-alpha induced phosphorylation of STAT3. The Janus family of tyrosine kinase (JAK) inhibitor I, an inhibitor of JAK 1, 2 and 3, attenuated TNF-alpha-induced phosphorylation of STAT3 and significantly reduced TNF-alpha-stimulated IL-6 release. Apocynin, an inhibitor of NADPH oxidase that suppresses intracellular reactive oxygen species, significantly suppressed TNF-alpha-induced IL-6 release and mRNA expression. However, apocynin failed to affect the phosphorylation of IkappaB, NFkappaB, p38 MAP kinase, SAPK/JNK or STAT3.
Conclusion: These results strongly suggest that TNF-alpha induces IL-6 synthesis through the JAK/STAT3 pathway in addition to p38 MAP kinase and SAPK/JNK in C6 glioma cells, and that phosphorylation of NFkappaB at Ser 536 and Ser 468, and NADPH oxidase are involved in TNF-alpha-stimulated IL-6 synthesis.
Figures






Similar articles
-
Mechanisms of interleukin-1beta-induced GDNF release from rat glioma cells.Brain Res. 2009 Jun 5;1274:11-20. doi: 10.1016/j.brainres.2009.03.063. Epub 2009 Apr 9. Brain Res. 2009. PMID: 19362079
-
Involvement of Rho-kinase in tumor necrosis factor-alpha-induced interleukin-6 release from C6 glioma cells.Neurochem Int. 2009 Nov;55(6):438-45. doi: 10.1016/j.neuint.2009.04.016. Epub 2009 May 7. Neurochem Int. 2009. PMID: 19427347
-
Midazolam suppresses interleukin-1β-induced interleukin-6 release from rat glial cells.J Neuroinflammation. 2011 Jun 17;8:68. doi: 10.1186/1742-2094-8-68. J Neuroinflammation. 2011. PMID: 21682888 Free PMC article.
-
Role of STAT3 in Genesis and Progression of Human Malignant Gliomas.Mol Neurobiol. 2017 Oct;54(8):5780-5797. doi: 10.1007/s12035-016-0103-0. Epub 2016 Sep 22. Mol Neurobiol. 2017. PMID: 27660268 Review.
-
Could signal transducer and activator of transcription 3 be a therapeutic target in obesity-related gastrointestinal malignancy?J Gastrointest Cancer. 2014 Mar;45(1):1-11. doi: 10.1007/s12029-013-9555-x. J Gastrointest Cancer. 2014. PMID: 24163144 Review.
Cited by
-
Cytokine patterns in brain tumour progression.Mediators Inflamm. 2013;2013:979748. doi: 10.1155/2013/979748. Epub 2013 Jun 24. Mediators Inflamm. 2013. PMID: 23864770 Free PMC article.
-
p38 MAPK inhibitors attenuate pro-inflammatory cytokine production and the invasiveness of human U251 glioblastoma cells.J Neurooncol. 2012 Aug;109(1):35-44. doi: 10.1007/s11060-012-0875-7. Epub 2012 Apr 19. J Neurooncol. 2012. PMID: 22528800
-
Astrocytoma development following complete multiple myeloma remission in a 49-year-old patient: A case report.Exp Ther Med. 2013 Aug;6(2):509-512. doi: 10.3892/etm.2013.1179. Epub 2013 Jun 25. Exp Ther Med. 2013. PMID: 24137217 Free PMC article.
-
IL-4, JAK-STAT signaling, and pain.JAKSTAT. 2013 Oct 1;2(4):e27638. doi: 10.4161/jkst.27638. Epub 2014 Jan 2. JAKSTAT. 2013. PMID: 24470980 Free PMC article. Review.
-
Neonatal Hyperoxia Downregulates Claudin-4, Occludin, and ZO-1 Expression in Rat Kidney Accompanied by Impaired Proximal Tubular Development.Oxid Med Cell Longev. 2020 Dec 2;2020:2641461. doi: 10.1155/2020/2641461. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 33343804 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

