Bright light activates a trigeminal nociceptive pathway
- PMID: 20206444
- PMCID: PMC2860692
- DOI: 10.1016/j.pain.2010.02.004
Bright light activates a trigeminal nociceptive pathway
Abstract
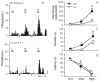
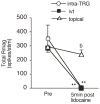
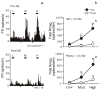
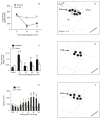
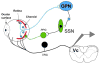
Bright light can cause ocular discomfort and/or pain; however, the mechanism linking luminance to trigeminal nerve activity is not known. In this study we identify a novel reflex circuit necessary for bright light to excite nociceptive neurons in superficial laminae of trigeminal subnucleus caudalis (Vc/C1). Vc/C1 neurons encoded light intensity and displayed a long delay (>10s) for activation. Microinjection of lidocaine into the eye or trigeminal root ganglion (TRG) inhibited light responses completely, whereas topical application onto the ocular surface had no effect. These findings indicated that light-evoked Vc/C1 activity was mediated by an intraocular mechanism and transmission through the TRG. Disrupting local vasomotor activity by intraocular microinjection of the vasoconstrictive agents, norepinephrine or phenylephrine, blocked light-evoked neural activity, whereas ocular surface or intra-TRG microinjection of norepinephrine had no effect. Pupillary muscle activity did not contribute since light-evoked responses were not altered by atropine. Microinjection of lidocaine into the superior salivatory nucleus diminished light-evoked Vc/C1 activity and lacrimation suggesting that increased parasympathetic outflow was critical for light-evoked responses. The reflex circuit also required input through accessory visual pathways since both Vc/C1 activity and lacrimation were prevented by local blockade of the olivary pretectal nucleus. These findings support the hypothesis that bright light activates trigeminal nerve activity through an intraocular mechanism driven by a luminance-responsive circuit and increased parasympathetic outflow to the eye.
Copyright 2010 International Association for the Study of Pain. Published by Elsevier B.V. All rights reserved.
Figures
Similar articles
-
Trigeminal interpolaris/caudalis transition neurons mediate reflex lacrimation evoked by bright light in the rat.Eur J Neurosci. 2012 Dec;36(11):3492-9. doi: 10.1111/j.1460-9568.2012.08272.x. Epub 2012 Sep 3. Eur J Neurosci. 2012. PMID: 22937868 Free PMC article.
-
Bright light produces Fos-positive neurons in caudal trigeminal brainstem.Neuroscience. 2009 Jun 2;160(4):858-64. doi: 10.1016/j.neuroscience.2009.03.003. Epub 2009 Mar 11. Neuroscience. 2009. PMID: 19285114
-
Trigeminal pathways for hypertonic saline- and light-evoked corneal reflexes.Neuroscience. 2014 Sep 26;277:716-23. doi: 10.1016/j.neuroscience.2014.07.052. Epub 2014 Jul 31. Neuroscience. 2014. PMID: 25086311 Free PMC article.
-
The effects of estrogen on temporomandibular joint pain as influenced by trigeminal caudalis neurons.J Oral Sci. 2020 Mar 28;62(2):150-155. doi: 10.2334/josnusd.19-0405. Epub 2020 Mar 4. J Oral Sci. 2020. PMID: 32132330 Review.
-
Non-neuronal cells act as crucial players in neuropathic orofacial pain.J Oral Biosci. 2024 Sep;66(3):491-495. doi: 10.1016/j.job.2024.07.005. Epub 2024 Jul 18. J Oral Biosci. 2024. PMID: 39032826 Review.
Cited by
-
Topical Tetrodotoxin Attenuates Photophobia Induced by Corneal Injury in the Rat.J Pain. 2015 Sep;16(9):881-6. doi: 10.1016/j.jpain.2015.06.002. Epub 2015 Jun 15. J Pain. 2015. PMID: 26086898 Free PMC article.
-
Migraine pathophysiology: anatomy of the trigeminovascular pathway and associated neurological symptoms, CSD, sensitization and modulation of pain.Pain. 2013 Dec;154 Suppl 1:10.1016/j.pain.2013.07.021. doi: 10.1016/j.pain.2013.07.021. Pain. 2013. PMID: 24347803 Free PMC article.
-
Investigation of light-induced lacrimation and pupillary responses in episodic migraine.PLoS One. 2020 Oct 30;15(10):e0241490. doi: 10.1371/journal.pone.0241490. eCollection 2020. PLoS One. 2020. PMID: 33125423 Free PMC article.
-
Cellular and physiological mechanisms underlying blood flow regulation in the retina and choroid in health and disease.Prog Retin Eye Res. 2012 Sep;31(5):377-406. doi: 10.1016/j.preteyeres.2012.04.004. Epub 2012 May 3. Prog Retin Eye Res. 2012. PMID: 22580107 Free PMC article. Review.
-
Correlation between higher-order aberration and photophobia after cataract surgery.PLoS One. 2022 Sep 15;17(9):e0274705. doi: 10.1371/journal.pone.0274705. eCollection 2022. PLoS One. 2022. PMID: 36107829 Free PMC article.
References
-
- Amini A, Digre K, Couldwell WT. Photophobia in a blind patient: An alternate visual pathway. Case report. J Neurosurg. 2006;105:765–768. - PubMed
-
- Belmonte C, Garcia-Hirschfeld J, Gallar J. Neurobiology of ocular pain. Progr Retinal Eye Res. 1997;16:117–156.
-
- Bereiter DA, Hargreaves KM, Hu JW. Trigeminal mechanisms of nociception: peripheral and brainstem organization. In: Basbaum A, Bushnell MC, editors. Science of Pain. Vol. 5. New York City: Elsevier; 2009. pp. 435–460.
-
- Bergua A, Schrodl F, Neuhuber WL. Vasoactive intestinal and calcitonin gene-related peptides, tyrosine hydroxylase and nitrergic markers in the innervation of the rat central retinal artery. Exp Eye Res. 2003;77:367–374. - PubMed
-
- Burstein R, Jakubowski M. Unitary hypothesis for multiple triggers of the pain and strain of migraine. J Comp Neurol. 2005;493:9–14. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

