Synthesis and structure-activity relationships of antimalarial 4-oxo-3-carboxyl quinolones
- PMID: 20206533
- PMCID: PMC2850272
- DOI: 10.1016/j.bmc.2010.02.013
Synthesis and structure-activity relationships of antimalarial 4-oxo-3-carboxyl quinolones
Abstract
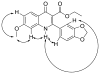
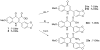
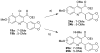
Malaria is endemic in tropical and subtropical regions of Africa, Asia, and the Americas. The increasing prevalence of multi-drug-resistant Plasmodium falciparum drives the ongoing need for the development of new antimalarial drugs. In this light, novel scaffolds to which the parasite has not been exposed are of particular interest. Recently, workers at the Swiss Tropical Institute discovered two novel 4-oxo-3-carboxyl quinolones active against the intra-erythrocytic stages of P. falciparum while carrying out rationally directed low-throughput screening of potential antimalarial agents as part of an effort directed by the World Health Organization. Here we report the design, synthesis, and preliminary pharmacologic characterization of a series of analogues of 4-oxo-3-carboxyl quinolones. These studies indicate that the series has good potential for preclinical development.
Copyright 2010 Elsevier Ltd. All rights reserved.
Figures




Similar articles
-
Synthesis, antimalarial activity, and structure-activity relationship of 7-(2-phenoxyethoxy)-4(1H)-quinolones.J Med Chem. 2011 Dec 22;54(24):8321-7. doi: 10.1021/jm200718m. Epub 2011 Nov 23. J Med Chem. 2011. PMID: 22111907 Free PMC article.
-
Endochin optimization: structure-activity and structure-property relationship studies of 3-substituted 2-methyl-4(1H)-quinolones with antimalarial activity.J Med Chem. 2010 Oct 14;53(19):7076-94. doi: 10.1021/jm1007903. J Med Chem. 2010. PMID: 20828199
-
Synthesis and activity of some quinolone derivatives against Plasmodium falciparum in vitro.Farmaco. 1996 Jun;51(6):407-12. Farmaco. 1996. PMID: 8766223
-
Role of Trifluoromethyl Substitution in Design of Antimalarial Quinolones: a Comprehensive Review.Top Curr Chem (Cham). 2019 Mar 5;377(2):9. doi: 10.1007/s41061-019-0234-7. Top Curr Chem (Cham). 2019. PMID: 30835005 Review.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
Cited by
-
Metal-free arylation of ethyl acetoacetate with hypervalent diaryliodonium salts: an immediate access to diverse 3-aryl-4(1H)-quinolones.J Org Chem. 2015 Mar 6;80(5):2513-2520. doi: 10.1021/jo5023958. Epub 2015 Feb 24. J Org Chem. 2015. PMID: 25558982 Free PMC article.
-
4(1H)-pyridone and 4(1H)-quinolone derivatives as antimalarials with erythrocytic, exoerythrocytic, and transmission blocking activities.Curr Top Med Chem. 2014;14(14):1693-705. doi: 10.2174/1568026614666140808124638. Curr Top Med Chem. 2014. PMID: 25116582 Free PMC article.
-
Synthesis, antimalarial activity, and structure-activity relationship of 7-(2-phenoxyethoxy)-4(1H)-quinolones.J Med Chem. 2011 Dec 22;54(24):8321-7. doi: 10.1021/jm200718m. Epub 2011 Nov 23. J Med Chem. 2011. PMID: 22111907 Free PMC article.
-
4-(1H)-Quinolones and 1,2,3,4-Tetrahydroacridin-9(10H)-ones prevent the transmission of Plasmodium falciparum to Anopheles freeborni.Antimicrob Agents Chemother. 2013 Dec;57(12):6187-95. doi: 10.1128/AAC.00492-13. Epub 2013 Sep 30. Antimicrob Agents Chemother. 2013. PMID: 24080648 Free PMC article.
-
New proapoptotic chemotherapeutic agents based on the quinolone-3-carboxamide scaffold acting by VEGFR-2 inhibition.Sci Rep. 2023 Jul 13;13(1):11346. doi: 10.1038/s41598-023-38264-w. Sci Rep. 2023. PMID: 37443185 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

