Histamine reduces flash sensitivity of on ganglion cells in the primate retina
- PMID: 20207974
- PMCID: PMC2904022
- DOI: 10.1167/iovs.09-4806
Histamine reduces flash sensitivity of on ganglion cells in the primate retina
Abstract
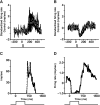
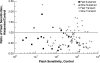
PURPOSE. In Old World primates, the retina receives input from histaminergic neurons in the posterior hypothalamus. They are a subset of the neurons that project throughout the central nervous system and fire maximally during the day. The contribution of these neurons to vision, was examined by applying histamine to a dark-adapted, superfused baboon eye cup preparation while making extracellular recordings from peripheral retinal ganglion cells. METHODS. The stimuli were 5-ms, 560-nm, weak, full-field flashes in the low scotopic range. Ganglion cells with sustained and transient ON responses and two cell types with OFF responses were distinguished; their responses were recorded with a 16-channel microelectrode array. RESULTS. Low micromolar doses of histamine decreased the rate of maintained firing and the light sensitivity of ON ganglion cells. Both sustained and transient ON cells responded similarly to histamine. There were no statistically significant effects of histamine in a more limited study of OFF ganglion cells. The response latencies of ON cells were approximately 5 ms slower, on average, when histamine was present. Histamine also reduced the signal-to-noise ratio of ON cells, particularly in those cells with a histamine-induced increase in maintained activity. CONCLUSIONS. A major action of histamine released from retinopetal axons under dark-adapted conditions, when rod signals dominate the response, is to reduce the sensitivity of ON ganglion cells to light flashes. These findings may relate to reports that humans are less sensitive to light stimuli in the scotopic range during the day, when histamine release in the retina is expected to be at its maximum.
Figures

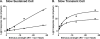


Similar articles
-
The effects of histamine on rat and monkey retinal ganglion cells.Vis Neurosci. 2004 Nov-Dec;21(6):935-43. doi: 10.1017/S0952523804216133. Vis Neurosci. 2004. PMID: 15733348
-
Light responses from one type of ON-OFF amacrine cells in the rabbit retina.J Neurophysiol. 1995 Dec;74(6):2460-8. doi: 10.1152/jn.1995.74.6.2460. J Neurophysiol. 1995. PMID: 8747206
-
Involvement of glycinergic neurons in the diminished surround activity of ganglion cells in the dark-adapted rabbit retina.Vis Neurosci. 1991 Jan;6(1):43-53. doi: 10.1017/s0952523800000894. Vis Neurosci. 1991. PMID: 1851036
-
Light-evoked increases in [K+]o in proximal portion of the dark-adapted cat retina.J Neurophysiol. 1989 Jun;61(6):1233-43. doi: 10.1152/jn.1989.61.6.1233. J Neurophysiol. 1989. PMID: 2746323
-
Retinopetal axons in mammals: emphasis on histamine and serotonin.Curr Eye Res. 2006 Jul-Aug;31(7-8):655-67. doi: 10.1080/02713680600776119. Curr Eye Res. 2006. PMID: 16877274 Free PMC article. Review.
Cited by
-
Novel Insight of Histamine and Its Receptor Ligands in Glaucoma and Retina Neuroprotection.Biomolecules. 2021 Aug 11;11(8):1186. doi: 10.3390/biom11081186. Biomolecules. 2021. PMID: 34439851 Free PMC article. Review.
-
Top-down modulation of the retinal code via histaminergic neurons of the hypothalamus.Sci Adv. 2024 Aug 30;10(35):eadk4062. doi: 10.1126/sciadv.adk4062. Epub 2024 Aug 28. Sci Adv. 2024. PMID: 39196935 Free PMC article.
-
The role of histamine in the retina: studies on the Hdc knockout mouse.PLoS One. 2014 Dec 29;9(12):e116025. doi: 10.1371/journal.pone.0116025. eCollection 2014. PLoS One. 2014. PMID: 25545149 Free PMC article.
-
Burning the candle at both ends: Intraretinal signaling of intrinsically photosensitive retinal ganglion cells.Front Cell Neurosci. 2023 Jan 6;16:1095787. doi: 10.3389/fncel.2022.1095787. eCollection 2022. Front Cell Neurosci. 2023. PMID: 36687522 Free PMC article. Review.
-
Metabolomic Study of a Rat Model of Retinal Detachment.Metabolites. 2022 Nov 7;12(11):1077. doi: 10.3390/metabo12111077. Metabolites. 2022. PMID: 36355160 Free PMC article.
References
-
- Reperant J, Ward R, Miceli D, et al. The centrifugal visual system of vertebrates: a comparative analysis of its functional anatomical organization. Brain Res Rev 2006;52:1–57 - PubMed
-
- Nowak JZ. Histamine in the retina and some other components of the visual system. Prog Retin Res 1993;12:41–74
-
- Labandeira-Garcia JL, Guerra-Seijas MJ, Gonzalez F, Perez R, Acuna C. Location of neurons projecting to the retina in mammals. Neurosci Res 1990;8:291–302 - PubMed
-
- Manning KA, Wilson JR, Uhlrich DJ. Histamine-immunoreactive neurons and their innervation of visual regions in the cortex, tectum, and thalamus in the primate Macaca mulatta. J Comp Neurol 1996;373:271–282 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

