Comparison of cobinamide to hydroxocobalamin in reversing cyanide physiologic effects in rabbits using diffuse optical spectroscopy monitoring
- PMID: 20210475
- PMCID: PMC2816993
- DOI: 10.1117/1.3290816
Comparison of cobinamide to hydroxocobalamin in reversing cyanide physiologic effects in rabbits using diffuse optical spectroscopy monitoring
Abstract
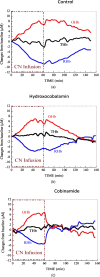
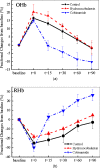
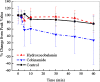
Our purpose is to compare cobinamide to hydroxocobalamin in reversing cyanide (CN)-induced physiologic effects in an animal model using diffuse optical spectroscopy (DOS). Cyanide poisoning is a major threat worldwide. Cobinamide is a novel molecule that can bind two molecules of cyanide, has a much higher binding affinity than hydroxocobalamin, and is more water soluble. We investigated the ability of equimolar doses of cobinamide and hydroxocobalamin to reverse the effects of cyanide exposure in an animal model monitored continuously by DOS. Cyanide toxicity was induced in 16 New Zealand white rabbits by intravenous infusion. Animals were divided into three groups: controls (n=5) received saline following cyanide, hydroxocobalamin (N=6) following cyanide, and cobinamide (N=5) following cyanide. Cobinamide caused significantly faster and more complete recovery of oxy- and deoxyhemoglobin concentrations in cyanide-exposed animals than hydroxocobalamin- or saline-treated animals, with a recovery time constant of 13.8+/-7.1 min compared to 75.4+/-25.1 and 76.4+/-42.7 min, for hydroxocobalamin- and saline-treated animals, respectively (p<0.0001). This study indicates that cobinamide more rapidly and completely reverses the physiologic effects of cyanide than equimolar doses of cobalamin at the dose used in this study, and CN effects and response can be followed noninvasively using DOS.
Figures
Similar articles
-
The vitamin B12 analog cobinamide is an effective hydrogen sulfide antidote in a lethal rabbit model.Clin Toxicol (Phila). 2014 Jun;52(5):490-7. doi: 10.3109/15563650.2014.904045. Epub 2014 Apr 9. Clin Toxicol (Phila). 2014. PMID: 24716792 Free PMC article.
-
Intramuscular cobinamide sulfite in a rabbit model of sublethal cyanide toxicity.Ann Emerg Med. 2010 Apr;55(4):352-63. doi: 10.1016/j.annemergmed.2009.12.002. Epub 2010 Jan 4. Ann Emerg Med. 2010. PMID: 20045579 Free PMC article.
-
Intravenous cobinamide versus hydroxocobalamin for acute treatment of severe cyanide poisoning in a swine (Sus scrofa) model.Ann Emerg Med. 2014 Dec;64(6):612-9. doi: 10.1016/j.annemergmed.2014.02.009. Epub 2014 Apr 18. Ann Emerg Med. 2014. PMID: 24746273 Free PMC article.
-
Hydroxocobalamin in cyanide poisoning.Clin Toxicol (Phila). 2012 Dec;50(10):875-85. doi: 10.3109/15563650.2012.742197. Epub 2012 Nov 19. Clin Toxicol (Phila). 2012. PMID: 23163594 Review.
-
Hydroxocobalamin as a cyanide antidote.Am J Ther. 2006 Mar-Apr;13(2):161-5. doi: 10.1097/01.mjt.0000174349.89671.8c. Am J Ther. 2006. PMID: 16645433 Review. No abstract available.
Cited by
-
Protection from cyanide-induced brain injury by the Nrf2 transcriptional activator carnosic acid.J Neurochem. 2015 Jun;133(6):898-908. doi: 10.1111/jnc.13074. Epub 2015 Mar 11. J Neurochem. 2015. PMID: 25692407 Free PMC article.
-
The vitamin B12 analog cobinamide is an effective hydrogen sulfide antidote in a lethal rabbit model.Clin Toxicol (Phila). 2014 Jun;52(5):490-7. doi: 10.3109/15563650.2014.904045. Epub 2014 Apr 9. Clin Toxicol (Phila). 2014. PMID: 24716792 Free PMC article.
-
Monitoring Dose Response of Cyanide Antidote Dimethyl Trisulfide in Rabbits Using Diffuse Optical Spectroscopy.J Med Toxicol. 2018 Dec;14(4):295-305. doi: 10.1007/s13181-018-0680-6. Epub 2018 Aug 9. J Med Toxicol. 2018. PMID: 30094773 Free PMC article.
-
A Review on Ingested Cyanide: Risks, Clinical Presentation, Diagnostics, and Treatment Challenges.J Med Toxicol. 2019 Apr;15(2):128-133. doi: 10.1007/s13181-018-0688-y. Epub 2018 Dec 11. J Med Toxicol. 2019. PMID: 30539383 Free PMC article. Review.
-
Cyanide inhibition and pyruvate-induced recovery of cytochrome c oxidase.J Bioenerg Biomembr. 2010 Oct;42(5):395-403. doi: 10.1007/s10863-010-9307-6. Epub 2010 Aug 20. J Bioenerg Biomembr. 2010. PMID: 20725851
References
-
- Baskin S. I. and Brewer T. G., “Medical aspects of chemical and biological warfare,” Chapter 10 in Textbook of Military Medicine Part I, Warfare, Weaponry, and the Casualty, Sidell F. R., Takafuji E. T., and Franz D. R., Eds., pp. 272–286, Borden Institute, Walter Reed Army Medical Center; Office of the Surgeon General, U.S. Army; U.S. Army Medical Dept. Center and School; U.S. Army Medical Research and Material Command; Uniformed Services University of the Health Sciences, Washington, DC: (1997).
-
- Strickland A., Wang R. Y., Hoffman R. S., and Goldfrank L. R., “Blood cyanide concentrations after smoke inhalation,” N. Engl. J. Med. NEJMAG 326(20), 1362 (1992). - PubMed
-
- Riddle K., “Hydrogen cyanide: fire smoke’s silent killer,” J. Emerg. Med. Serv. (JEMS) 29(8), suppl. 5 (2004). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

