Neuroprotective effects of the drug GVT (monosodium luminol) are mediated by the stabilization of Nrf2 in astrocytes
- PMID: 20211212
- PMCID: PMC4109344
- DOI: 10.1016/j.neuint.2010.02.017
Neuroprotective effects of the drug GVT (monosodium luminol) are mediated by the stabilization of Nrf2 in astrocytes
Abstract
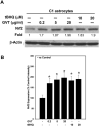
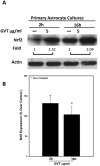
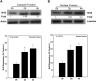
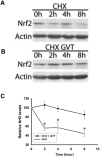
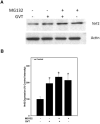
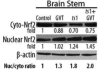
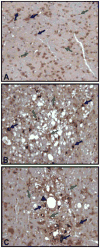
Oxidative stress is implicated in various kinds of neurological disorders, including human immunodeficiency virus (HIV) associated dementia (HAD). Our laboratory has been studying the murine retrovirus ts1, a pathogenic mutant of the Moloney murine leukemia virus (MoMuLV), as a model for HAD. Like HIV in humans, ts1 induces oxidative stress and progressive neurodegeneration in mice. We have shown previously that an antioxidant and anti-inflammatory drug GVT or MSL (monosodium luminol) suppresses ts1-induced oxidative stress, attenuates the development of spongiform encephalopathy, and delays hind limb paralysis in infected mice. It is known that upregulation of the nuclear transcription factor NF-E2-related factor 2 (Nrf2) is involved in upregulating cellular antioxidant defenses. Since Nrf2 is associated with elevation of antioxidant defenses in general, and since GVT suppresses ts1-induced neurodegeneration, our aim in this study was to determine whether GVT neuroprotection is linked to Nrf2 upregulation in the brain. We report here that GVT upregulates the levels of Nrf2, both in primary astrocyte cultures and in brainstem of ts1-infected mice. Significant upregulation of Nrf2 expression by GVT occurs in both the cytosolic and nuclear fractions of cultured astrocytes and brainstem cells. Notably, although GVT treatment increases Nrf2 protein levels in cultured astrocytes and brainstem tissues, Nrf2 mRNA levels are not altered. This suggests that the neuroprotective effects of GVT may be mediated by the stabilization of the Nrf2 protein, allowing continuous upregulation of Nrf2 levels in the astrocytes.
Copyright 2010 Elsevier Ltd. All rights reserved.
Figures
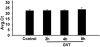
Similar articles
-
Retrovirus-induced oxidative stress with neuroimmunodegeneration is suppressed by antioxidant treatment with a refined monosodium alpha-luminol (Galavit).J Virol. 2006 May;80(9):4557-69. doi: 10.1128/JVI.80.9.4557-4569.2006. J Virol. 2006. PMID: 16611916 Free PMC article.
-
Astrocytes survive chronic infection and cytopathic effects of the ts1 mutant of the retrovirus Moloney murine leukemia virus by upregulation of antioxidant defenses.J Virol. 2006 Apr;80(7):3273-84. doi: 10.1128/JVI.80.7.3273-3284.2006. J Virol. 2006. PMID: 16537594 Free PMC article.
-
The drug monosodium luminol (GVT) preserves crypt-villus epithelial organization and allows survival of intestinal T cells in mice infected with the ts1 retrovirus.Immunol Lett. 2009 Feb 21;122(2):150-8. doi: 10.1016/j.imlet.2008.12.012. Epub 2009 Jan 30. Immunol Lett. 2009. PMID: 19186189
-
The Nrf2-ARE pathway: an indicator and modulator of oxidative stress in neurodegeneration.Ann N Y Acad Sci. 2008 Dec;1147:61-9. doi: 10.1196/annals.1427.036. Ann N Y Acad Sci. 2008. PMID: 19076431 Free PMC article. Review.
-
The role of modulation of antioxidant enzyme systems in the treatment of neurodegenerative diseases.J Enzyme Inhib Med Chem. 2016;31(sup3):194-204. doi: 10.1080/14756366.2016.1205047. Epub 2016 Jul 7. J Enzyme Inhib Med Chem. 2016. PMID: 27389167 Review.
Cited by
-
Behavioral and histological assessment of a novel treatment of neuroHIV in humanized mice.Res Sq [Preprint]. 2023 Dec 13:rs.3.rs-3678629. doi: 10.21203/rs.3.rs-3678629/v1. Res Sq. 2023. PMID: 38168407 Free PMC article. Preprint.
-
TLR2 activation induces antioxidant defence in human monocyte-macrophage cell line models.Oncotarget. 2017 Apr 21;8(33):54243-54264. doi: 10.18632/oncotarget.17342. eCollection 2017 Aug 15. Oncotarget. 2017. PMID: 28903338 Free PMC article.
-
Phenylbutyric acid suppresses protein accumulation-mediated ER stress in retrovirus-infected astrocytes and delays onset of paralysis in infected mice.Neurochem Int. 2010 Dec;57(7):738-48. doi: 10.1016/j.neuint.2010.08.010. Epub 2010 Sep 8. Neurochem Int. 2010. PMID: 20813146 Free PMC article.
-
Activation of the nuclear factor E2-related factor 2/antioxidant response element pathway is neuroprotective after spinal cord injury.J Neurotrauma. 2012 Mar 20;29(5):936-45. doi: 10.1089/neu.2011.1922. Epub 2011 Sep 21. J Neurotrauma. 2012. PMID: 21806470 Free PMC article.
-
Maysin and Its Flavonoid Derivative from Centipedegrass Attenuates Amyloid Plaques by Inducting Humoral Immune Response with Th2 Skewed Cytokine Response in the Tg (APPswe, PS1dE9) Alzheimer's Mouse Model.PLoS One. 2017 Jan 10;12(1):e0169509. doi: 10.1371/journal.pone.0169509. eCollection 2017. PLoS One. 2017. PMID: 28072821 Free PMC article.
References
-
- Betteridge DJ. What is oxidative stress? Metabolism. 2000;49:3–8. - PubMed
-
- Cerimele F, Battle T, Lynch R, Frank DA, Murad E, Cohen C, Macaron N, Sixbey J, Smith K, Watnick RS, Eliopoulos A, Shehata B, Arbiser JL. Reactive oxygen signaling and MAPK activation distinguish Epstein-Barr Virus (EBV)-positive versus EBV-negative Burkitt's lymphoma. Proc Natl Acad Sci U S A. 2005;102:175–179. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

