Regulation of Kv channel expression and neuronal excitability in rat medial nucleus of the trapezoid body maintained in organotypic culture
- PMID: 20211981
- PMCID: PMC2876802
- DOI: 10.1113/jphysiol.2009.186676
Regulation of Kv channel expression and neuronal excitability in rat medial nucleus of the trapezoid body maintained in organotypic culture
Abstract
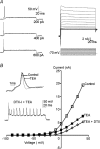
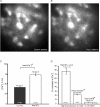
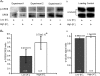
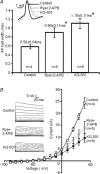
Principal neurons of the medial nucleus of the trapezoid body (MNTB) express a spectrum of voltage-dependent K(+) conductances mediated by Kv1-Kv4 channels, which shape action potential (AP) firing and regulate intrinsic excitability. Postsynaptic factors influencing expression of Kv channels were explored using organotypic cultures of brainstem prepared from P9-P12 rats and maintained in either low (5 mm, low-K) or high (25 mm, high-K) [K(+)](o) medium. Whole cell patch-clamp recordings were made after 7-28 days in vitro. MNTB neurons cultured in high-K medium maintained a single AP firing phenotype, while low-K cultures had smaller K(+) currents, enhanced excitability and fired multiple APs. The calyx of Held inputs degenerated within 3 days in culture, having lost their major afferent input; this preparation of calyx-free MNTB neurons allowed the effects of postsynaptic depolarisation to be studied with minimal synaptic activity. The depolarization caused by the high-K aCSF only transiently increased spontaneous AP firing (<2 min) and did not measurably increase synaptic activity. Chronic depolarization in high-K cultures raised basal levels of [Ca(2+)](i), increased Kv3 currents and shortened AP half-widths. These events relied on raised [Ca(2+)](i), mediated by influx through voltage-gated calcium channels (VGCCs) and release from intracellular stores, causing an increase in cAMP-response element binding protein (CREB) phosphorylation. Block of VGCCs or of CREB function suppressed Kv3 currents, increased AP duration, and reduced Kv3.3 and c-fos expression. Real-time PCR revealed higher Kv3.3 and Kv1.1 mRNA in high-K compared to low-K cultures, although the increased Kv1.1 mRNA was mediated by a CREB-independent mechanism. We conclude that Kv channel expression and hence the intrinsic membrane properties of MNTB neurons are homeostatically regulated by [Ca(2+)](i)-dependent mechanisms and influenced by sustained depolarization of the resting membrane potential.
Figures




Comment in
-
Controlling auditory excitability: the benefits of a cultured environment.J Physiol. 2010 May 1;588(Pt 9):1387-8. doi: 10.1113/jphysiol.2010.189712. J Physiol. 2010. PMID: 20436041 Free PMC article. No abstract available.
References
-
- Berridge MJ. Neuronal calcium signalling. Neuron. 1998;21:13–26. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

