The H3K27me3 demethylase dUTX is a suppressor of Notch- and Rb-dependent tumors in Drosophila
- PMID: 20212086
- PMCID: PMC2863695
- DOI: 10.1128/MCB.01633-09
The H3K27me3 demethylase dUTX is a suppressor of Notch- and Rb-dependent tumors in Drosophila
Abstract
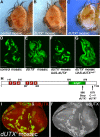
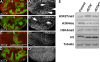
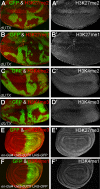
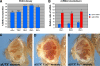
Trimethylated lysine 27 of histone H3 (H3K27me3) is an epigenetic mark for gene silencing and can be demethylated by the JmjC domain of UTX. Excessive H3K27me3 levels can cause tumorigenesis, but little is known about the mechanisms leading to those cancers. Mutants of the Drosophila H3K27me3 demethylase dUTX display some characteristics of Trithorax group mutants and have increased H3K27me3 levels in vivo. Surprisingly, dUTX mutations also affect H3K4me1 levels in a JmjC-independent manner. We show that a disruption of the JmjC domain of dUTX results in a growth advantage for mutant cells over adjacent wild-type tissue due to increased proliferation. The growth advantage of dUTX mutant tissue is caused, at least in part, by increased Notch activity, demonstrating that dUTX is a Notch antagonist. Furthermore, the inactivation of Retinoblastoma (Rbf in Drosophila) contributes to the growth advantage of dUTX mutant tissue. The excessive activation of Notch in dUTX mutant cells leads to tumor-like growth in an Rbf-dependent manner. In summary, these data suggest that dUTX is a suppressor of Notch- and Rbf-dependent tumors in Drosophila melanogaster and may provide a model for UTX-dependent tumorigenesis in humans.
Figures


References
-
- Agger, K., P. A. Cloos, J. Christensen, D. Pasini, S. Rose, J. Rappsilber, I. Issaeva, E. Canaani, A. E. Salcini, and K. Helin. 2007. UTX and JMJD3 are histone H3K27 demethylases involved in HOX gene regulation and development. Nature 449:731-734. - PubMed
-
- Bagchi, S., R. Weinmann, and P. Raychaudhuri. 1991. The retinoblastoma protein copurifies with E2F-I, an E1A-regulated inhibitor of the transcription factor E2F. Cell 65:1063-1072. - PubMed
-
- Baonza, A., and M. Freeman. 2005. Control of cell proliferation in the Drosophila eye by Notch signaling. Dev. Cell 8:529-539. - PubMed
-
- Blatch, G. L., and M. Lassle. 1999. The tetratricopeptide repeat: a structural motif mediating protein-protein interactions. Bioessays 21:932-939. - PubMed
-
- Boyer, L. A., K. Plath, J. Zeitlinger, T. Brambrink, L. A. Medeiros, T. I. Lee, S. S. Levine, M. Wernig, A. Tajonar, M. K. Ray, G. W. Bell, A. P. Otte, M. Vidal, D. K. Gifford, R. A. Young, and R. Jaenisch. 2006. Polycomb complexes repress developmental regulators in murine embryonic stem cells. Nature 441:349-353. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- GM074977/GM/NIGMS NIH HHS/United States
- R01 GM068016/GM/NIGMS NIH HHS/United States
- GM61672/GM/NIGMS NIH HHS/United States
- R01 GM081543/GM/NIGMS NIH HHS/United States
- R01 CA089455/CA/NCI NIH HHS/United States
- CA089455/CA/NCI NIH HHS/United States
- GM069905/GM/NIGMS NIH HHS/United States
- GM081543/GM/NIGMS NIH HHS/United States
- GM068016/GM/NIGMS NIH HHS/United States
- R01 CA150265/CA/NCI NIH HHS/United States
- R01 GM074977/GM/NIGMS NIH HHS/United States
- R01 GM069905/GM/NIGMS NIH HHS/United States
- R01 GM061672/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
