Dynamics within the CD95 death-inducing signaling complex decide life and death of cells
- PMID: 20212524
- PMCID: PMC2858442
- DOI: 10.1038/msb.2010.6
Dynamics within the CD95 death-inducing signaling complex decide life and death of cells
Abstract
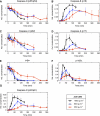
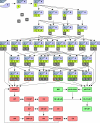
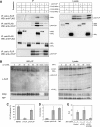
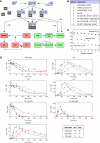
This study explores the dilemma in cellular signaling that triggering of CD95 (Fas/APO-1) in some situations results in cell death and in others leads to the activation of NF-kappaB. We established an integrated kinetic mathematical model for CD95-mediated apoptotic and NF-kappaB signaling. Systematic model reduction resulted in a surprisingly simple model well approximating experimentally observed dynamics. The model postulates a new link between c-FLIP(L) cleavage in the death-inducing signaling complex (DISC) and the NF-kappaB pathway. We validated experimentally that CD95 stimulation resulted in an interaction of p43-FLIP with the IKK complex followed by its activation. Furthermore, we showed that the apoptotic and NF-kappaB pathways diverge already at the DISC. Model and experimental analysis of DISC formation showed that a subtle balance of c-FLIP(L) and procaspase-8 determines life/death decisions in a nonlinear manner. We present an integrated model describing the complex dynamics of CD95-mediated apoptosis and NF-kappaB signaling.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures


Similar articles
-
Stoichiometry of the CD95 death-inducing signaling complex: experimental and modeling evidence for a death effector domain chain model.Mol Cell. 2012 Jul 27;47(2):306-19. doi: 10.1016/j.molcel.2012.05.006. Epub 2012 Jun 7. Mol Cell. 2012. PMID: 22683265
-
Rapid up-regulation of c-FLIP expression by BCR signaling through the PI3K/Akt pathway inhibits simultaneously induced Fas-mediated apoptosis in murine B lymphocytes.Immunol Lett. 2007 Mar 15;109(1):36-46. doi: 10.1016/j.imlet.2006.12.009. Epub 2007 Jan 22. Immunol Lett. 2007. PMID: 17275920
-
Human T-cell leukemia virus type-I oncoprotein Tax inhibits Fas-mediated apoptosis by inducing cellular FLIP through activation of NF-kappaB.Genes Cells. 2006 Feb;11(2):177-91. doi: 10.1111/j.1365-2443.2006.00927.x. Genes Cells. 2006. PMID: 16436054
-
Regulation of CD95/Fas signaling at the DISC.Cell Death Differ. 2012 Jan;19(1):36-41. doi: 10.1038/cdd.2011.155. Epub 2011 Nov 11. Cell Death Differ. 2012. PMID: 22075988 Free PMC article. Review.
-
Cellular FLICE-like inhibitory proteins (c-FLIPs): fine-tuners of life and death decisions.Exp Cell Res. 2012 Jul 1;318(11):1324-31. doi: 10.1016/j.yexcr.2012.01.019. Epub 2012 Jan 28. Exp Cell Res. 2012. PMID: 22309778 Review.
Cited by
-
Effects of Periostracum Cicadae on Cytokines and Apoptosis Regulatory Proteins in an IgA Nephropathy Rat Model.Int J Mol Sci. 2018 May 29;19(6):1599. doi: 10.3390/ijms19061599. Int J Mol Sci. 2018. PMID: 29844269 Free PMC article.
-
Caspase-8 activity has an essential role in CD95/Fas-mediated MAPK activation.Cell Death Dis. 2011 Oct 6;2(10):e212. doi: 10.1038/cddis.2011.93. Cell Death Dis. 2011. PMID: 21975294 Free PMC article.
-
Generalized logical model based on network topology to capture the dynamical trends of cellular signaling pathways.BMC Syst Biol. 2016 Jan 11;10 Suppl 1(Suppl 1):7. doi: 10.1186/s12918-015-0249-9. BMC Syst Biol. 2016. PMID: 26818802 Free PMC article.
-
p53-mediated delayed NF-κB activity enhances etoposide-induced cell death in medulloblastoma.Cell Death Dis. 2010 May 13;1(5):e41. doi: 10.1038/cddis.2010.16. Cell Death Dis. 2010. PMID: 21364648 Free PMC article.
-
Modulating cell-to-cell variability and sensitivity to death ligands by co-drugging.Phys Biol. 2013 Jun;10(3):035002. doi: 10.1088/1478-3975/10/3/035002. Epub 2013 Jun 4. Phys Biol. 2013. PMID: 23735516 Free PMC article.
References
-
- Arnold R, Liou J, Drexler HC, Weiss A, Kiefer F (2001) Caspase-mediated cleavage of hematopoietic progenitor kinase 1 (HPK1) converts an activator of NFkappaB into an inhibitor of NFkappaB. J Biol Chem 276: 14675–14684 - PubMed
-
- Ashkenazi A, Dixit VM (1998) Death receptors: signaling and modulation. Science 281: 1305–1308 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

