Murine visceral leishmaniasis: IgM and polyclonal B-cell activation lead to disease exacerbation
- PMID: 20213734
- PMCID: PMC2944234
- DOI: 10.1002/eji.200939455
Murine visceral leishmaniasis: IgM and polyclonal B-cell activation lead to disease exacerbation
Abstract
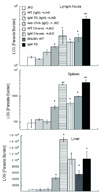
In visceral leishmaniasis, the draining LN (DLN) is the initial site for colonization and establishment of infection after intradermal transmission by the sand fly vector; however, little is known about the developing immune response within this site. Using an intradermal infection model, which allows for parasite visceralization, we have examined the ongoing immune responses in the DLN of BALB/c mice infected with Leishmania infantum. Although not unexpected, at early times post-infection there is a marked B-cell expansion in the DLN, which persists throughout infection. However, the characteristics of this response were of interest; as early as day 7 post-infection, polyclonal antibodies (TNP, OVA, chromatin) were observed and the levels appeared comparable to the specific anti-leishmania response. Although B-cell-deficient JhD BALB/c mice are relatively resistant to infection, neither B-cell-derived IL-10 nor B-cell antigen presentation appear to be primarily responsible for the elevated parasitemia. However, passive transfer and reconstitution of JhD BALB/c with secretory immunoglobulins, (IgM or IgG; specific or non-specific immune complexes) results in increased susceptibility to L. infantum infection. Further, JhD BALB/c mice transgenetically reconstituted to secrete IgM demonstrated exacerbated disease in comparison to WT BALB/c mice as early as 2 days post-infection. Evidence suggests that complement activation (generation of C5a) and signaling via the C5a receptor (CD88) is related to the disease exacerbation caused by IgM rather than cytokine levels (IL-10 or IFN-gamma). Overall these studies indicate that polyclonal B-cell activation, which is known to be associated with human visceral leishmaniasis, is an early and intrinsic characteristic of disease and may represent a target for therapeutic intervention.
Conflict of interest statement
Figures
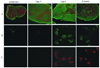

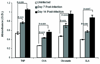
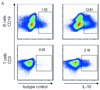

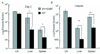
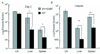

Similar articles
-
Leishmania infantum amastin protein incorporated in distinct adjuvant systems induces protection against visceral leishmaniasis.Cytokine. 2020 May;129:155031. doi: 10.1016/j.cyto.2020.155031. Epub 2020 Feb 12. Cytokine. 2020. PMID: 32062145
-
Investigations on the effects of anti-Leishmania major serum on the progression of Leishmania infantum infection in vivo and in vitro - implications of heterologous exposure to Leishmania spp.Parasitol Res. 2021 May;120(5):1771-1780. doi: 10.1007/s00436-021-07130-x. Epub 2021 Apr 1. Parasitol Res. 2021. PMID: 33792813
-
Understanding Resistance vs. Susceptibility in Visceral Leishmaniasis Using Mouse Models of Leishmania infantum Infection.Front Cell Infect Microbiol. 2019 Mar 1;9:30. doi: 10.3389/fcimb.2019.00030. eCollection 2019. Front Cell Infect Microbiol. 2019. PMID: 30881923 Free PMC article.
-
Influence of the host and parasite strain in a mouse model of visceral Leishmania infantum infection.FEMS Immunol Med Microbiol. 1998 Jul;21(3):231-9. doi: 10.1111/j.1574-695X.1998.tb01170.x. FEMS Immunol Med Microbiol. 1998. PMID: 9718213
-
B-1 B cell IgM antibody initiates T cell elicitation of contact sensitivity.Curr Top Microbiol Immunol. 2000;252:171-7. doi: 10.1007/978-3-642-57284-5_18. Curr Top Microbiol Immunol. 2000. PMID: 11125474 Review.
Cited by
-
How to B(e)-1 Important Cell During Leishmania Infection.Front Cell Infect Microbiol. 2020 Jan 14;9:424. doi: 10.3389/fcimb.2019.00424. eCollection 2019. Front Cell Infect Microbiol. 2020. PMID: 31993374 Free PMC article. Review.
-
IgG1 is pathogenic in Leishmania mexicana infection.J Immunol. 2010 Dec 1;185(11):6939-46. doi: 10.4049/jimmunol.1002484. Epub 2010 Oct 29. J Immunol. 2010. PMID: 21037092 Free PMC article.
-
B cell: T cell interactions occur within hepatic granulomas during experimental visceral leishmaniasis.PLoS One. 2012;7(3):e34143. doi: 10.1371/journal.pone.0034143. Epub 2012 Mar 30. PLoS One. 2012. PMID: 22479545 Free PMC article.
-
Visualizing the In Vivo Dynamics of Anti-Leishmania Immunity: Discoveries and Challenges.Front Immunol. 2021 May 19;12:671582. doi: 10.3389/fimmu.2021.671582. eCollection 2021. Front Immunol. 2021. PMID: 34093571 Free PMC article. Review.
-
IL-4 contributes to failure, and colludes with IL-10 to exacerbate Leishmania donovani infection following administration of a subcutaneous leishmanial antigen vaccine.BMC Microbiol. 2014 Jan 15;14:8. doi: 10.1186/1471-2180-14-8. BMC Microbiol. 2014. PMID: 24428931 Free PMC article.
References
-
- Stanley AC, Engwerda CR. Balancing immunity and pathology in visceral leishmaniasis. Immunol Cell Biol. 2007;85:138–147. - PubMed
-
- Kaye PM, Svensson M, Ato M, Maroof A, Polley R, Stager S, Zubairi S, Engwerda CR. The immunopathology of experimental visceral leishmaniasis. Immunol Rev. 2004;201:239–253. - PubMed
-
- Mueller SN, Ahmed R. Lymphoid stroma in the initiation and control of immune responses. Immunol Rev. 2008;224:284–294. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R21 AI027811/AI/NIAID NIH HHS/United States
- T32 AI07404/AI/NIAID NIH HHS/United States
- R01 AI027811/AI/NIAID NIH HHS/United States
- AI27811/AI/NIAID NIH HHS/United States
- R01 GM062134/GM/NIGMS NIH HHS/United States
- T32 AI007404/AI/NIAID NIH HHS/United States
- R01 AI045044/AI/NIAID NIH HHS/United States
- AI45044/AI/NIAID NIH HHS/United States
- R56 AI043603/AI/NIAID NIH HHS/United States
- AI-068730/AI/NIAID NIH HHS/United States
- AI-43603/AI/NIAID NIH HHS/United States
- GM-62134/GM/NIGMS NIH HHS/United States
- P01 AI068730/AI/NIAID NIH HHS/United States
- R01 AI043603/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

