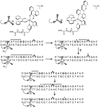
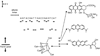
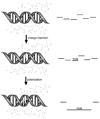
Mechanisms for DNA charge transport
- PMID: 20214403
- PMCID: PMC2879062
- DOI: 10.1021/cr900228f
Mechanisms for DNA charge transport
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

