Approaches to phase 1 clinical trial design focused on safety, efficiency, and selected patient populations: a report from the clinical trial design task force of the national cancer institute investigational drug steering committee
- PMID: 20215542
- PMCID: PMC5207802
- DOI: 10.1158/1078-0432.CCR-09-1961
Approaches to phase 1 clinical trial design focused on safety, efficiency, and selected patient populations: a report from the clinical trial design task force of the national cancer institute investigational drug steering committee
Abstract
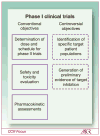
The goals and objectives of phase 1 clinical trials are changing to include further evaluation of endpoints such as molecular targeted effects, in addition to dose-toxicity profile of the investigational agent. Because of these changes in focus, the National Cancer Institute and Investigational Drug Steering Committee's Task Force on Clinical Trial Design met to evaluate the most efficient ways to design and implement early clinical trials with novel therapeutics. Clinical approaches discussed included the conventional 3 + 3 cohort expansion phase 1 design, multi-institutional phase 1 studies, accelerated titration designs, continual reassessment methods, the study of specific target patient populations, and phase 0 studies. Each of these approaches uniquely contributes to some aspect of the phase 1 study, with all focused on dose and schedule determination, patient safety, and limited patient exposure to ineffective doses of investigational agent. The benefit of labor-intensive generation of preliminary biomarker evidence of target inhibition, as well as the value of molecular profiling of the study population, is considered. New drug development is expensive and the failure rate remains high. By identifying patient populations expected to respond to the study agent and tailoring the treatment with a novel drug, investigators will be one step closer to personalizing cancer treatment. The "fail early and fast" approach is acceptable if the appropriate patient population is evaluated in the phase 1 trial. The approaches outlined in this overview address the merits, advantages, disadvantages, and obstacles encountered during first in human studies.
Figures


References
-
- Von Hoff DD, Turner J. Response rates, duration of response, and dose response effects in phase I studies of antineoplastics. Invest New Drugs. 1991;9:115–22. - PubMed
-
- Le Tourneau C, et al. Choice of Starting Dose of Molecularly Targeted Agents Evaluated in First-in-Human Phase I Cancer Clinical Trials. J Clin Oncol. 2010 In press. - PubMed
-
- Eisenhauer EA, O’Dwyer PJ, Christian M, et al. Phase I clinical trial design in cancer drug development. J Clin Oncol. 2000;18:684–92. - PubMed
-
- Dowlati A, Manda S, Gibbons J, et al. Multi-institutional phase I trials of anticancer agents. J Clin Oncol. 2008;26:1926–31. - PubMed
-
- Parulekar WR, Eisenhauer EA. Phase I trial design for solid tumor studies of targeted, non-cytotoxic agents: theory and practice. J Natl Cancer Inst. 2004;96:990–7. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

