Stress-activated cap'n'collar transcription factors in aging and human disease
- PMID: 20215646
- PMCID: PMC2991085
- DOI: 10.1126/scisignal.3112re3
Stress-activated cap'n'collar transcription factors in aging and human disease
Abstract
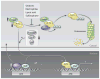
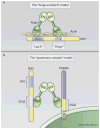
Cap'n'collar (Cnc) transcription factors are conserved in metazoans and have important developmental and homeostatic functions. The vertebrate Nrf1, Nrf2, and Nrf3; the Caenorhabditis elegans SKN-1; and the Drosophila CncC comprise a subgroup of Cnc factors that mediate adaptive responses to cellular stress. The most studied stress-activated Cnc factor is Nrf2, which orchestrates the transcriptional response of cells to oxidative stressors and electrophilic xenobiotics. In rodent models, signaling by Nrf2 defends against oxidative stress and aging-associated disorders, such as neurodegeneration, respiratory diseases, and cancer. In humans, polymorphisms that decrease Nrf2 abundance have been associated with various pathologies of the skin, respiratory system, and digestive tract. In addition to preventing disease in rodents and humans, Cnc factors have life-span-extending and anti-aging functions in invertebrates. However, despite the pro-longevity and antioxidant roles of stress-activated Cnc factors, their activity paradoxically declines in aging model organisms and in humans suffering from progressive respiratory disease or neurodegeneration. We review the roles and regulation of stress-activated Cnc factors across species, present all reported instances in which their activity is paradoxically decreased in aging and disease, and discuss the possibility that the pharmacological restoration of Nrf2 signaling may be useful in the prevention and treatment of age-related diseases.
Conflict of interest statement
Figures

References
-
- Bowerman B, Eaton BA, Priess JR. skn-1, a maternally expressed gene required to specify the fate of ventral blastomeres in the early C. elegans embryo. Cell. 1992;68:1061–1075. - PubMed
-
- Mohler J, Vani K, Leung S, Epstein A. Segmentally restricted, cephalic expression of a leucine zipper gene during Drosophila embryogenesis. Mech Dev. 1991;34:3–9. - PubMed
-
- Chan JY, Cheung MC, Moi P, Chan K, Kan YW. Chromosomal localization of the human NF-E2 family of bZIP transcription factors by fluorescence in situ hybridization. Hum Genet. 1995;95:265–269. - PubMed
-
- Andrews NC, Erdjument-Bromage H, Davidson MB, Tempst P, Orkin SH. Erythroid transcription factor NF-E2 is a haematopoietic-specific basic-leucine zipper protein. Nature. 1993;362:722–728. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

