N,N-dimethyl leucines as novel isobaric tandem mass tags for quantitative proteomics and peptidomics
- PMID: 20218596
- PMCID: PMC2859709
- DOI: 10.1021/ac902778d
N,N-dimethyl leucines as novel isobaric tandem mass tags for quantitative proteomics and peptidomics
Abstract
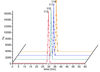
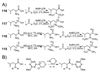
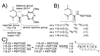
Herein, we describe the development and application of a set of novel N,N-dimethyl leucine (DiLeu) 4-plex isobaric tandem mass (MS(2)) tagging reagents with high quantitation efficacy and greatly reduced cost for neuropeptide and protein analysis. DiLeu reagents serve as attractive alternatives for isobaric tags for relative and absolute quantitation (iTRAQ) and tandem mass tags (TMTs) due to their synthetic simplicity, labeling efficiency, and improved fragmentation efficiency. DiLeu reagent resembles the general structure of a tandem mass tag in that it contains an amine reactive group (triazine ester) targeting the N-terminus and epsilon-amino group of the lysine side chain of a peptide, a balance group, and a reporter group. A mass shift of 145.1 Da is observed for each incorporated label. Intense a(1) reporter ions at m/z 115.1, 116.1, 117.1, and 118.1 are observed for all pooled samples upon MS(2). All labeling reagents are readily synthesized from commercially available chemicals with greatly reduced cost. Labels 117 and 118 can be synthesized in one step and labels 115 and 116 can be synthesized in two steps. Both DiLeu and iTRAQ reagents show comparable protein sequence coverage (approximately 43%) and quantitation accuracy (<15%) for tryptically digested protein samples. Furthermore, enhanced fragmentation of DiLeu labeling reagents offers greater confidence in protein identification and neuropeptide sequencing from complex neuroendocrine tissue extracts from a marine model organism, Callinectes sapidus.
Figures





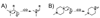
Similar articles
-
Development and characterization of novel 8-plex DiLeu isobaric labels for quantitative proteomics and peptidomics.Rapid Commun Mass Spectrom. 2015 Jun 30;29(12):1115-24. doi: 10.1002/rcm.7201. Rapid Commun Mass Spectrom. 2015. PMID: 25981542 Free PMC article.
-
High-Throughput Quantitative Proteomics Enabled by Mass Defect-Based 12-Plex DiLeu Isobaric Tags.Methods Mol Biol. 2016;1410:169-94. doi: 10.1007/978-1-4939-3524-6_10. Methods Mol Biol. 2016. PMID: 26867744
-
21-plex DiLeu Isobaric Tags for High-Throughput Quantitative Proteomics.Anal Chem. 2020 Jun 16;92(12):8228-8234. doi: 10.1021/acs.analchem.0c00473. Epub 2020 May 28. Anal Chem. 2020. PMID: 32401496 Free PMC article.
-
Trends in the Design of New Isobaric Labeling Reagents for Quantitative Proteomics.Molecules. 2019 Feb 15;24(4):701. doi: 10.3390/molecules24040701. Molecules. 2019. PMID: 30781343 Free PMC article. Review.
-
Multi-functional MBIT for peptide tandem mass spectrometry.Mass Spectrom Rev. 2015 Mar-Apr;34(2):209-18. doi: 10.1002/mas.21435. Epub 2014 May 28. Mass Spectrom Rev. 2015. PMID: 24872020 Review.
Cited by
-
Development and characterization of novel 8-plex DiLeu isobaric labels for quantitative proteomics and peptidomics.Rapid Commun Mass Spectrom. 2015 Jun 30;29(12):1115-24. doi: 10.1002/rcm.7201. Rapid Commun Mass Spectrom. 2015. PMID: 25981542 Free PMC article.
-
Capillary Electrophoresis-Electrospray Ionization-Mass Spectrometry for Quantitative Analysis of Glycans Labeled with Multiplex Carbonyl-Reactive Tandem Mass Tags.Anal Chem. 2015 Jul 7;87(13):6527-34. doi: 10.1021/acs.analchem.5b01835. Epub 2015 Jun 11. Anal Chem. 2015. PMID: 25981625 Free PMC article.
-
Clinical Proteomics in Colorectal Cancer, a Promising Tool for Improving Personalised Medicine.Proteomes. 2018 Dec 2;6(4):49. doi: 10.3390/proteomes6040049. Proteomes. 2018. PMID: 30513835 Free PMC article. Review.
-
Recent Advances in Understanding of Alzheimer's Disease Progression through Mass Spectrometry-Based Metabolomics.Phenomics. 2022 Feb;2(1):1-17. doi: 10.1007/s43657-021-00036-9. Epub 2022 Feb 22. Phenomics. 2022. PMID: 35656096 Free PMC article.
-
Boost-DiLeu: Enhanced Isobaric N,N-Dimethyl Leucine Tagging Strategy for a Comprehensive Quantitative Glycoproteomic Analysis.Anal Chem. 2022 Aug 30;94(34):11773-11782. doi: 10.1021/acs.analchem.2c01773. Epub 2022 Aug 12. Anal Chem. 2022. PMID: 35960654 Free PMC article.
References
-
- Ong S-E, Mann M. Nat Chem Biol. 2005;1:252–262. - PubMed
-
- Gygi SP, Rist B, Gerber SA, Turecek F, Gelb MH, Aebersold R. Nature Biotechnology. 1999;17:994. - PubMed
-
- Li J, Steen H, Gygi SP. Mol Cell Proteomics. 2003;2:1198–1204. - PubMed
-
- Hansen KC, Schmitt-Ulms G, Chalkley RJ, Hirsch J, Baldwin MA, Burlingame AL. Mol Cell Proteomics. 2003;2:299–314. - PubMed
-
- Ong S-E, Blagoev B, Kratchmarova I, Kristensen DB, Steen H, Pandey A, Mann M. Mol Cell Proteomics. 2002;1:376–386. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

