Role of human immunodeficiency virus type 1 integrase in uncoating of the viral core
- PMID: 20219923
- PMCID: PMC2863833
- DOI: 10.1128/JVI.02382-09
Role of human immunodeficiency virus type 1 integrase in uncoating of the viral core
Abstract
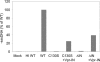
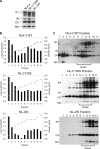
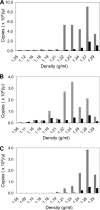
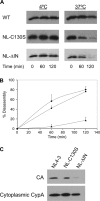
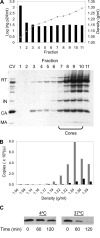
After membrane fusion with a target cell, the core of human immunodeficiency virus type 1 (HIV-1) enters into the cytoplasm, where uncoating occurs. The cone-shaped core is composed of the viral capsid protein (CA), which disassembles during uncoating. The underlying factors and mechanisms governing uncoating are poorly understood. Several CA mutations can cause changes in core stability and a block at reverse transcription, demonstrating the requirement for optimal core stability during viral replication. HIV-1 integrase (IN) catalyzes the insertion of the viral cDNA into the host genome, and certain IN mutations are pleiotropic. Similar to some CA mutants, two IN mutants, one with a complete deletion of IN (NL-DeltaIN) and the other with a Cys-to-Ser substitution (NL-C130S), were noninfectious, with a replication block at reverse transcription. Compared to the wild type (WT), the cytoplasmic CA levels of the IN mutants in infected cells were reduced, suggesting accelerated uncoating. The role of IN during uncoating was examined by isolating and characterizing cores from NL-DeltaIN and NL-C130S. Both IN mutants could form functional cores, but the core yield and stability were decreased. Also, virion incorporation of cyclophilin A (CypA), a cellular peptidyl-prolyl isomerase that binds specifically to CA, was decreased in the IN mutants. Cores isolated from WT virus depleted of CypA had an unstable-core phenotype, confirming a role of CypA in promoting optimal core stability. Taken together, our results indicate that IN is required during uncoating for maintaining CypA-CA interaction, which promotes optimal stability of the viral core.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

