Release and elementary mechanisms of nitric oxide in hair cells
- PMID: 20220083
- PMCID: PMC2867581
- DOI: 10.1152/jn.00017.2010
Release and elementary mechanisms of nitric oxide in hair cells
Abstract
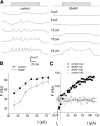
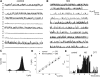
The enzyme nitric oxide (NO) synthase, that produces the signaling molecule NO, has been identified in several cell types in the inner ear. However, it is unclear whether a measurable quantity of NO is released in the inner ear to confer specific functions. Indeed, the functional significance of NO and the elementary cellular mechanism thereof are most uncertain. Here, we demonstrate that the sensory epithelia of the frog saccule release NO and explore its release mechanisms by using self-referencing NO-selective electrodes. Additionally, we investigated the functional effects of NO on electrical properties of hair cells and determined their underlying cellular mechanism. We show detectable amounts of NO are released by hair cells (>50 nM). Furthermore, a hair-cell efferent modulator acetylcholine produces at least a threefold increase in NO release. NO not only attenuated the baseline membrane oscillations but it also increased the magnitude of current required to generate the characteristic membrane potential oscillations. This resulted in a rightward shift in the frequency-current relationship and altered the excitability of hair cells. Our data suggest that these effects ensue because NO reduces whole cell Ca(2+) current and drastically decreases the open probability of single-channel events of the L-type and non L-type Ca(2+) channels in hair cells, an effect that is mediated through direct nitrosylation of the channel and activation of protein kinase G. Finally, NO increases the magnitude of Ca(2+)-activated K(+) currents via direct NO nitrosylation. We conclude that NO-mediated inhibition serves as a component of efferent nerve modulation of hair cells.
Figures







Similar articles
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Null mutation of alpha1D Ca2+ channel gene results in deafness but no vestibular defect in mice.J Assoc Res Otolaryngol. 2004 Jun;5(2):215-26. doi: 10.1007/s10162-003-4020-3. J Assoc Res Otolaryngol. 2004. PMID: 15357422 Free PMC article.
-
Direct measurement of single-channel Ca(2+) currents in bullfrog hair cells reveals two distinct channel subtypes.J Physiol. 2001 Aug 1;534(Pt 3):669-89. doi: 10.1111/j.1469-7793.2001.00669.x. J Physiol. 2001. PMID: 11483699 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Drugs for preventing postoperative nausea and vomiting in adults after general anaesthesia: a network meta-analysis.Cochrane Database Syst Rev. 2020 Oct 19;10(10):CD012859. doi: 10.1002/14651858.CD012859.pub2. Cochrane Database Syst Rev. 2020. PMID: 33075160 Free PMC article.
Cited by
-
Electrophysiological properties of vestibular hair cells isolated from human crista.Front Neurol. 2025 Jan 22;15:1501914. doi: 10.3389/fneur.2024.1501914. eCollection 2024. Front Neurol. 2025. PMID: 39911744 Free PMC article.
-
Nitrergic modulation of ion channel function in regulating neuronal excitability.Channels (Austin). 2021 Dec;15(1):666-679. doi: 10.1080/19336950.2021.2002594. Channels (Austin). 2021. PMID: 34802368 Free PMC article.
-
Efferent synaptic transmission at the vestibular type II hair cell synapse.J Neurophysiol. 2020 Aug 1;124(2):360-374. doi: 10.1152/jn.00143.2020. Epub 2020 Jul 1. J Neurophysiol. 2020. PMID: 32609559 Free PMC article.
-
Equal sensitivity of Cav1.2 and Cav1.3 channels to the opposing modulations of PKA and PKG in mouse chromaffin cells.J Physiol. 2012 Oct 15;590(20):5053-73. doi: 10.1113/jphysiol.2012.236729. Epub 2012 Jul 23. J Physiol. 2012. PMID: 22826131 Free PMC article.
-
The amplitude and inactivation properties of the delayed potassium currents are regulated by protein kinase activity in hair cells of the frog semicircular canals.PLoS One. 2013 Jul 2;8(7):e67784. doi: 10.1371/journal.pone.0067784. Print 2013. PLoS One. 2013. PMID: 23844092 Free PMC article.
References
-
- Ahmmed GU, Xu Y, Hong Dong P, Zhang Z, Eiserich J, Chiamvimonvat N. Nitric oxide modulates cardiac Na(+) channel via protein kinase A and protein kinase G. Circ Res 89: 1005–1013, 2001. - PubMed
-
- Almanza A, Navarrete F, Vega R, Soto E. Modulation of voltage-gated Ca2+ current in vestibular hair cells by nitric oxide. J Neurophysiol 97: 1188–1195, 2007. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

