Reliability of antimalarial sensitivity tests depends on drug mechanisms of action
- PMID: 20220159
- PMCID: PMC2863886
- DOI: 10.1128/JCM.02250-09
Reliability of antimalarial sensitivity tests depends on drug mechanisms of action
Abstract
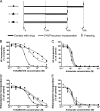
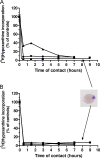
In vitro antimalarial activity tests play a pivotal role in malaria drug research or for monitoring drug resistance in field isolates. We applied two isotopic tests, two enzyme-linked immunosorbent assays (ELISA) and the SYBR green I fluorescence-based assay, to test artesunate and chloroquine, the metabolic inhibitors atovaquone and pyrimethamine, our fast-acting choline analog T3/SAR97276, and doxycycline, which has a delayed death profile. Isotopic tests based on hypoxanthine and ethanolamine incorporation are the most reliable tests provided when they are applied after one full 48-h parasite cycle. The SYBR green assay, which measures the DNA content, usually requires 72 h of incubation to obtain reliable results. When delayed death is suspected, specific protocols are required with increasing incubation times up to 96 h. In contrast, both ELISA tests used (pLDH and HRP2) appear to be problematic, leading to disappointing and even erroneous results for molecules that do not share an artesunatelike profile. The reliability of these tests is linked to the mode of action of the drug, and the conditions required to get informative results are hard to predict. Our results suggest some minimal conditions to apply these tests that should give rise to a standard 50% inhibitory concentration, regardless of the mechanism of action of the compounds, and highlight that the most commonly used in vitro antimalarial activity tests do not have the same potential. Some of them might not detect the antimalarial potential of new classes of compounds with innovative modes of action, which subsequently could become promising new antimalarial drugs.
Figures
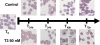
References
-
- Ancelin, M. L., M. Calas, J. Bompart, G. Cordina, D. Martin, M. Ben Bari, T. Jei, P. Druilhe, and H. J. Vial. 1998. Antimalarial activity of 77 phospholipid polar head analogs: close correlation between inhibition of phospholipid metabolism and in vitro Plasmodium falciparum growth. Blood 91:1426-1437. - PubMed
-
- Bacon, D. J., C. Latour, C. Lucas, O. Colina, P. Ringwald, and S. Picot. 2007. Comparison of a SYBR green I-based assay with a histidine-rich protein II enzyme-linked immunosorbent assay for in vitro antimalarial drug efficacy testing and application to clinical isolates. Antimicrob. Agents Chemother. 51:1172-1178. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

