Oncolytic herpes simplex virus armed with xenogeneic homologue of prostatic acid phosphatase enhances antitumor efficacy in prostate cancer
- PMID: 20220784
- PMCID: PMC3243306
- DOI: 10.1038/gt.2010.20
Oncolytic herpes simplex virus armed with xenogeneic homologue of prostatic acid phosphatase enhances antitumor efficacy in prostate cancer
Abstract
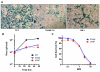
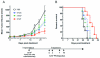
Prostate cancer is one of the most prevalent cancers in men. Replication-competent oncolytic herpes simplex virus (oHSV) vectors are a powerful antitumor therapy that can exert at least two effects: direct cytocidal activity that selectively kills cancer cells and induction of antitumor immunity. In addition, oHSV vectors can also function as a platform to deliver transgenes of interest. In these studies, we have examined the expression of a xenogeneic homologue of the prostate cancer antigen, prostatic acid phosphatase (PAP), with the goal of enhancing virotherapy against PAP-expressing tumors. PAP has already been used for cancer vaccination in patients with prostate cancer. Here we show that treatment with oHSV bPDelta6 expressing xenogeneic human PAP (hPAP) significantly reduces tumor growth and increases survival of C57/BL6 mice bearing mouse TRAMP-C2 prostate tumors, whereas expression of syngeneic mouse PAP (mPAP) from the same oHSV vector did not enhance antitumor activity. Treatment of mice bearing metastatic TRAMP-C2 lung tumors with oHSV-expressing hPAP resulted in fewer tumor nodules. To our knowledge, this is the first report of oncolytic viruses being used to express xenoantigens. These data lend support to the concept of combining oncolytic and immunogenic therapies as a way to improve therapy of metastatic prostate cancer.
Figures

Similar articles
-
Enhanced therapeutic efficacy of IL-12, but not GM-CSF, expressing oncolytic herpes simplex virus for transgenic mouse derived prostate cancers.Cancer Gene Ther. 2006 Mar;13(3):253-65. doi: 10.1038/sj.cgt.7700900. Cancer Gene Ther. 2006. PMID: 16179929
-
Combination of vinblastine and oncolytic herpes simplex virus vector expressing IL-12 therapy increases antitumor and antiangiogenic effects in prostate cancer models.Cancer Gene Ther. 2013 Jan;20(1):17-24. doi: 10.1038/cgt.2012.75. Epub 2012 Nov 9. Cancer Gene Ther. 2013. PMID: 23138870 Free PMC article.
-
IL-12 Expressing oncolytic herpes simplex virus promotes anti-tumor activity and immunologic control of metastatic ovarian cancer in mice.J Ovarian Res. 2016 Oct 27;9(1):70. doi: 10.1186/s13048-016-0282-3. J Ovarian Res. 2016. PMID: 27784340 Free PMC article.
-
Oncolytic virus therapy using genetically engineered herpes simplex viruses.Front Biosci. 2008 Jan 1;13:2060-4. doi: 10.2741/2823. Front Biosci. 2008. PMID: 17981691 Review.
-
Oncolytic Herpes Simplex Virus-Based Therapies for Cancer.Cells. 2021 Jun 18;10(6):1541. doi: 10.3390/cells10061541. Cells. 2021. PMID: 34207386 Free PMC article. Review.
Cited by
-
Oncolytic virotherapy using herpes simplex virus: how far have we come?Oncolytic Virother. 2015 Nov 25;4:207-19. doi: 10.2147/OV.S66086. eCollection 2015. Oncolytic Virother. 2015. PMID: 27512683 Free PMC article. Review.
-
Taking a Stab at Cancer; Oncolytic Virus-Mediated Anti-Cancer Vaccination Strategies.Biomedicines. 2017 Jan 4;5(1):3. doi: 10.3390/biomedicines5010003. Biomedicines. 2017. PMID: 28536346 Free PMC article. Review.
-
Anti-tumor effect of oncolytic herpes simplex virus G47delta on human nasopharyngeal carcinoma.Chin J Cancer. 2011 Dec;30(12):831-41. doi: 10.5732/cjc.011.10301. Epub 2011 Nov 4. Chin J Cancer. 2011. PMID: 22059912 Free PMC article.
-
Oncolytic viruses and their application to cancer immunotherapy.Cancer Immunol Res. 2014 Apr;2(4):295-300. doi: 10.1158/2326-6066.CIR-14-0015. Cancer Immunol Res. 2014. PMID: 24764576 Free PMC article. Review.
-
Sharpening the Edge for Precision Cancer Immunotherapy: Targeting Tumor Antigens through Oncolytic Vaccines.Front Immunol. 2017 Jul 13;8:800. doi: 10.3389/fimmu.2017.00800. eCollection 2017. Front Immunol. 2017. PMID: 28751892 Free PMC article. Review.
References
-
- Moul JW, Mouraviev V, Sun L, Schroeck FR, Polascik TJ. Prostate cancer: the new landscape. Curr Opin Urol. 2009;19:154–160. - PubMed
-
- Mitchell RE, Chang SS. Current controversies in the treatment of high-risk prostate cancer. Curr Opin Urol. 2008;18:263–8. - PubMed
-
- Sia M, Rosewall T, Warde P. Radiotherapy as primary treatment modality. Front Radiat Ther Oncol. 2008;41:15–25. - PubMed
-
- Martuza RL, Malick A, Markert JM, Ruffner KL, Coen DM. Experimental therapy of human glioma by means of a genetically engineered virus mutant. Science. 1991;252:854–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

