Maternal immunization with ovalbumin prevents neonatal allergy development and up-regulates inhibitory receptor Fc gamma RIIB expression on B cells
- PMID: 20222978
- PMCID: PMC2848204
- DOI: 10.1186/1471-2172-11-11
Maternal immunization with ovalbumin prevents neonatal allergy development and up-regulates inhibitory receptor Fc gamma RIIB expression on B cells
Abstract
Background: Preconception allergen immunization prevents neonatal allergen sensitization in mice by a complex interaction between regulatory cells/factors and antibodies. The present study assessed the influence of maternal immunization with ovalbumin (OVA) on the immune response of 3 day-old and 3 week-old offspring immunized or non-immunized with OVA and evaluated the effect of IgG treatment during fetal development or neonatal period.
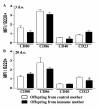
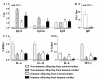
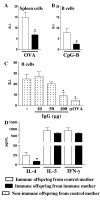
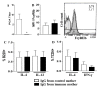
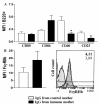
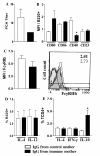
Results: Maternal immunization with OVA showed increased levels of Fc gamma RIIb expression in splenic B cells of neonates, which were maintained for up to 3 weeks and not affected by additional postnatal OVA immunization. Maternal immunization also exerted a down-modulatory effect on both IL-4 and IFN-gamma-secreting T cells and IL-4 and IL-12- secreting B cells. Furthermore, immunized neonates from immunized mothers showed a marked inhibition of antigen-specific IgE Ab production and lowered Th2/Th1 cytokine levels, whereas displaying enhanced Fc gamma RIIb expression on B cells. These offspring also showed reduced antigen-specific proliferative response and lowered B cell responsiveness. Moreover, in vitro evaluation revealed an impairment of B cell activation upon engagement of B cell antigen receptor by IgG from OVA-immunized mice. Finally, in vivo IgG transference during pregnancy or breastfeeding revealed that maternal Ab transference was able to increase regulatory cytokines, such as IL-10, in the prenatal stage; yet only the postnatal treatment prevented neonatal sensitization. None of the IgG treatments induced immunological changes in the offspring, as it was observed for those from OVA-immunized mothers.
Conclusion: Maternal immunization upregulates the inhibitory Fc gamma RIIb expression on offspring B cells, avoiding skewed Th2 response and development of allergy. These findings contribute to the advancement of prophylactic strategies to prevent allergic diseases in early life.
Figures

Similar articles
-
Maternal-fetal interaction: preconception immunization in mice prevents neonatal sensitization induced by allergen exposure during pregnancy and breastfeeding.Immunology. 2007 Sep;122(1):107-15. doi: 10.1111/j.1365-2567.2007.02618.x. Epub 2007 Jul 3. Immunology. 2007. PMID: 17608811 Free PMC article.
-
Tolerogenic microenvironment in neonatal period induced by maternal immunization with ovalbumin.Immunobiology. 2014 May;219(5):377-84. doi: 10.1016/j.imbio.2014.01.002. Epub 2014 Jan 10. Immunobiology. 2014. PMID: 24582301
-
Early life interventions to prevent allergy in the offspring: the role of maternal immunization and postnatal mucosal allergen exposure.Int Arch Allergy Immunol. 2012;158(3):261-75. doi: 10.1159/000332963. Epub 2012 Mar 2. Int Arch Allergy Immunol. 2012. PMID: 22398405
-
Therapeutic interference with interferon-gamma (IFN-gamma) and soluble IL-4 receptor (sIL-4R) in allergic diseases.Behring Inst Mitt. 1995 Jun;(96):118-30. Behring Inst Mitt. 1995. PMID: 7575347 Review.
-
IL-4 from Th2-type cells suppresses induction of delayed-type hypersensitivity elicited shortly after immunization.Immunol Cell Biol. 2003 Dec;81(6):424-30. doi: 10.1046/j.1440-1711.2003.01194.x. Immunol Cell Biol. 2003. PMID: 14636239 Review.
Cited by
-
Maternal allergic disease history affects childhood allergy development through impairment of neonatal regulatory T-cells.Respir Res. 2016 Sep 20;17(1):114. doi: 10.1186/s12931-016-0430-8. Respir Res. 2016. PMID: 27646403 Free PMC article.
-
Mechanism underlying polyvalent IgG-induced regulatory T cell activation and its clinical application: Anti-idiotypic regulatory T cell theory for immune tolerance.Front Immunol. 2023 Nov 29;14:1242860. doi: 10.3389/fimmu.2023.1242860. eCollection 2023. Front Immunol. 2023. PMID: 38094290 Free PMC article. Review.
-
Maternal Immunization: New Perspectives on Its Application Against Non-Infectious Related Diseases in Newborns.Vaccines (Basel). 2017 Aug 1;5(3):20. doi: 10.3390/vaccines5030020. Vaccines (Basel). 2017. PMID: 28763018 Free PMC article. Review.
-
Inter- and transgenerational epigenetic inheritance: evidence in asthma and COPD?Clin Epigenetics. 2015 May 1;7(1):53. doi: 10.1186/s13148-015-0085-1. eCollection 2015. Clin Epigenetics. 2015. PMID: 26052354 Free PMC article.
-
Allergen-specific IgG as a mediator of allergy inhibition: Lessons from mother to child.Hum Vaccin Immunother. 2017 Mar 4;13(3):507-513. doi: 10.1080/21645515.2016.1244592. Epub 2016 Nov 3. Hum Vaccin Immunother. 2017. PMID: 27808600 Free PMC article. Review.
References
-
- Seeger M, Thierse HJ, Lange H, Shaw L, Hansen H, Lemke H. Antigen-independent suppression of the IgE immune response to bee venom phospholipase A(2) by maternally derived monoclonal IgG antibodies. Eur J Immunol. 1998;28:2124–2130. doi: 10.1002/(SICI)1521-4141(199807)28:07<2124::AID-IMMU2124>3.0.CO;2-A. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

