Improved killing of human high-grade glioma cells by combining ionizing radiation with oncolytic parvovirus H-1 infection
- PMID: 20224643
- PMCID: PMC2833303
- DOI: 10.1155/2010/350748
Improved killing of human high-grade glioma cells by combining ionizing radiation with oncolytic parvovirus H-1 infection
Abstract
Purpose: To elucidate the influence of ionizing radiation (IR) on the oncolytic activity of Parvovirus H-1 (H-1PV) in human high-grade glioma cells.
Methods: Short term cultures of human high-grade gliomas were irradiated at different doses and infected with H-1PV. Cell viability was assessed by determining relative numbers of surviving cells. Replication of H-1PV was measured by RT-PCR of viral RNA, fluorescence-activated cell sorter (FACS) analysis and the synthesis of infectious virus particles. To identify a possible mechanism for radiation induced change in the oncolytic activity of H-1PV we performed cell cycle analyses.
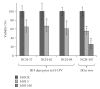
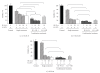
Results: Previous irradiation rendered glioma cells fully permissive to H-1PV infection. Irradiation 24 hours prior to H-1PV infection led to increased cell killing most notably in radioresistant glioma cells. Intracellular levels of NS-1, the main effector of H-1PV induced cytotoxicity, were elevated after irradiation. S-phase levels were increased one day after irradiation improving S-phase dependent viral replication and cytotoxicity.
Conclusion: This study demonstrates intact susceptibility of previously irradiated glioma-cells for H-1PV induced oncolysis. The combination of ionizing radiation followed by H-1PV infection increased viral cytotoxicity, especially in radioresistant gliomas. These findings support the ongoing development of a clinical trial of H-1PV in patients with recurrent glioblastomas.
Figures



Similar articles
-
Therapeutic implications of the enhanced short and long-term cytotoxicity of radiation treatment followed by oncolytic parvovirus H-1 infection in high-grade glioma cells.Bioeng Bugs. 2010 Nov-Dec;1(6):429-33. doi: 10.4161/bbug.1.6.12943. Bioeng Bugs. 2010. PMID: 21468212 Free PMC article.
-
Regression of glioma in rat models by intranasal application of parvovirus h-1.Clin Cancer Res. 2011 Aug 15;17(16):5333-42. doi: 10.1158/1078-0432.CCR-10-3124. Epub 2011 Jun 29. Clin Cancer Res. 2011. PMID: 21715567
-
Combined oncolytic and vaccination activities of parvovirus H-1 in a metastatic tumor model.Oncol Rep. 2007 Jun;17(6):1493-9. Oncol Rep. 2007. PMID: 17487410
-
Double-faceted mechanism of parvoviral oncosuppression.Curr Opin Virol. 2015 Aug;13:17-24. doi: 10.1016/j.coviro.2015.03.008. Epub 2015 Apr 2. Curr Opin Virol. 2015. PMID: 25841215 Review.
-
H-1 Parvovirus as a Cancer-Killing Agent: Past, Present, and Future.Viruses. 2019 Jun 18;11(6):562. doi: 10.3390/v11060562. Viruses. 2019. PMID: 31216641 Free PMC article. Review.
Cited by
-
Virus-Based Immunotherapy of Glioblastoma.Cancers (Basel). 2019 Feb 5;11(2):186. doi: 10.3390/cancers11020186. Cancers (Basel). 2019. PMID: 30764570 Free PMC article. Review.
-
Oncolytic Viruses to Treat Ovarian Cancer Patients - a Review of Results From Clinical Trials.Geburtshilfe Frauenheilkd. 2012 Feb;72(2):132-136. doi: 10.1055/s-0031-1298281. Geburtshilfe Frauenheilkd. 2012. PMID: 25374430 Free PMC article.
-
In vitro Characterization of Enhanced Human Immune Responses by GM-CSF Encoding HSV-1-Induced Melanoma Cells.Onco Targets Ther. 2022 Oct 27;15:1291-1307. doi: 10.2147/OTT.S350136. eCollection 2022. Onco Targets Ther. 2022. PMID: 36310770 Free PMC article.
-
Novel Therapies in Glioblastoma Treatment: Review of Glioblastoma; Current Treatment Options; and Novel Oncolytic Viral Therapies.Med Sci (Basel). 2023 Dec 23;12(1):1. doi: 10.3390/medsci12010001. Med Sci (Basel). 2023. PMID: 38249077 Free PMC article. Review.
-
The Oncolytic Virotherapy Era in Cancer Management: Prospects of Applying H-1 Parvovirus to Treat Blood and Solid Cancers.Front Oncol. 2017 May 12;7:93. doi: 10.3389/fonc.2017.00093. eCollection 2017. Front Oncol. 2017. PMID: 28553616 Free PMC article.
References
-
- Maher EA, Furnari FB, Bachoo RM, et al. Malignant glioma: genetics and biology of a grave matter. Genes and Development. 2001;15(11):1311–1333. - PubMed
-
- Zhu Y, Parada LF. The molecular and genetic basis of neurological tumours. Nature Reviews Cancer. 2002;2(8):616–626. - PubMed
-
- DeAngelis LM. Chemotherapy for brain tumors—a new beginning. The New England Journal of Medicine. 2005;352(10):1036–1038. - PubMed
-
- Stupp R, Dietrich P-Y, Kraljevic SO, et al. Promising survival for patients with newly diagnosed glioblastoma multiforme treated with concomitant radiation plus temozolomide followed by adjuvant temozolomide. Journal of Clinical Oncology. 2002;20(5):1375–1382. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

