N-terminally fusion of Her2/neu to HSP70 decreases efficiency of Her2/neu DNA vaccine
- PMID: 20224916
- PMCID: PMC3006617
- DOI: 10.1007/s12192-010-0175-0
N-terminally fusion of Her2/neu to HSP70 decreases efficiency of Her2/neu DNA vaccine
Abstract
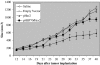
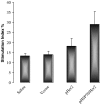
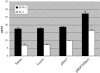
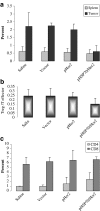
DNA vaccines consisted of tumor-associated antigen (TAA) are well suited for immunotherapy against tumor. The construct can contain TAA fused to an appropriate molecule (biologic adjuvant) to improve the efficacy of anti-tumor immune response. Heat shock protein 70 (HSP70) has been shown to be an excellent candidate, capable of cross-priming TAA by antigen presenting cells leading to a robust T-cell response. However, the relationship between strong T-cell responses and tumor rejection is not always mutually exclusive, for which TAA loss or activation of suppressive mechanisms may occur. HSP70 fused to downstream of Her2/neu as DNA vaccine has been shown to be efficient against Her2-expressing tumors. In this study, we examined if N-terminally fusion of Her2/neu to HSP70 could also improve efficiency of Her2/neu DNA vaccine. Therefore, mice with an established Her2/neu expressing tumor were immunized with DNA vaccine consisting of extracellular and trans-membrane domain (EC+TM) of rat Her2/neu alone or N-terminally fused to HSP70 and immune response was evaluated. Administration of rat Her2/neu led to partial control of tumor progression. Surprisingly, fusion of HSP70 to N-terminal of rat Her2/neu led to tumor progression. Our result proposes that fusion direction of biologic adjuvant is an important consideration when Her2/neu is used.
Figures

Similar articles
-
Adjuvant activity of GP96 C-terminal domain towards Her2/neu DNA vaccine is fusion direction-dependent.Cell Stress Chaperones. 2011 Jan;16(1):41-8. doi: 10.1007/s12192-010-0219-5. Epub 2010 Aug 22. Cell Stress Chaperones. 2011. PMID: 20730610 Free PMC article.
-
Comparison of adjuvant activity of N- and C-terminal domain of gp96 in a Her2-positive breast cancer model.Cell Stress Chaperones. 2011 Jul;16(4):449-57. doi: 10.1007/s12192-011-0258-6. Epub 2011 Feb 26. Cell Stress Chaperones. 2011. PMID: 21359667 Free PMC article.
-
A novel vaccine strategy to induce mycobacterial antigen-specific Th1 responses by utilizing the C-terminal domain of heat shock protein 70.FEMS Immunol Med Microbiol. 2011 Mar;61(2):189-96. doi: 10.1111/j.1574-695X.2010.00762.x. Epub 2011 Jan 18. FEMS Immunol Med Microbiol. 2011. PMID: 21204994
-
Targeting of the non-mutated tumor antigen HER2/neu to mature dendritic cells induces an integrated immune response that protects against breast cancer in mice.Breast Cancer Res. 2012 Mar 7;14(2):R39. doi: 10.1186/bcr3135. Breast Cancer Res. 2012. PMID: 22397502 Free PMC article.
-
Efficient induction of a Her2-specific anti-tumor response by dendritic cells pulsed with a Hsp70L1-Her2(341-456) fusion protein.Cell Mol Immunol. 2011 Sep;8(5):424-32. doi: 10.1038/cmi.2011.21. Epub 2011 Jul 25. Cell Mol Immunol. 2011. PMID: 21785448 Free PMC article.
Cited by
-
Adjuvant activity of GP96 C-terminal domain towards Her2/neu DNA vaccine is fusion direction-dependent.Cell Stress Chaperones. 2011 Jan;16(1):41-8. doi: 10.1007/s12192-010-0219-5. Epub 2010 Aug 22. Cell Stress Chaperones. 2011. PMID: 20730610 Free PMC article.
-
The significance of heat shock proteins in breast cancer therapy.Med Oncol. 2013;30(2):575. doi: 10.1007/s12032-013-0575-y. Epub 2013 Apr 20. Med Oncol. 2013. PMID: 23606238 Review.
-
Comparison of adjuvant activity of N- and C-terminal domain of gp96 in a Her2-positive breast cancer model.Cell Stress Chaperones. 2011 Jul;16(4):449-57. doi: 10.1007/s12192-011-0258-6. Epub 2011 Feb 26. Cell Stress Chaperones. 2011. PMID: 21359667 Free PMC article.
-
Mini-chaperones: potential immuno-stimulators in vaccine design.Hum Vaccin Immunother. 2013 Jan;9(1):153-61. doi: 10.4161/hv.22248. Epub 2012 Oct 29. Hum Vaccin Immunother. 2013. PMID: 23108356 Free PMC article. Review.
-
Co-administration of GP96 and Her2/neu DNA vaccine in a Her2 breast cancer model.Cell Stress Chaperones. 2010 Nov;15(6):977-84. doi: 10.1007/s12192-010-0208-8. Epub 2010 Jun 12. Cell Stress Chaperones. 2010. PMID: 20544406 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

