A hormone-activated central pattern generator for courtship
- PMID: 20226670
- PMCID: PMC2858972
- DOI: 10.1016/j.cub.2010.02.027
A hormone-activated central pattern generator for courtship
Abstract
Background: Medicinal leeches (Hirudo spp.) are simultaneous hermaphrodites. Mating occurs after a stereotyped twisting and oral exploration that result in the alignment of the male and/or female gonopores of one leech with the complementary gonopores of a partner. The neural basis of this behavior is presently unknown and currently impossible to study directly because electrophysiological recording techniques disrupt the behavior.
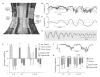
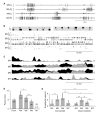
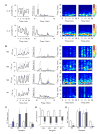
Results: Here we report that (Arg(8))-conopressin G and two other members of the oxytocin/vasopressin family of peptide hormones induce in Hirudo verbana a sequence of behaviors that closely mimic elements of spontaneous reproductive behavior. Through a series of progressively more reduced preparations, we show that one of these behaviors, a stereotyped twisting that is instrumental in aligning gonopores in preparation for copulation, is the product of a central pattern generator that consists of oscillators in ganglia M5 and M6 (the ganglia in the reproductive segments of the leech), and also in ganglion M4, which was not previously known to play a role in reproductive behavior. We find that the behavior is periodic, with a remarkably long cycle period of around five minutes, placing it among the slowest behavioral rhythms (other than diurnal and annual rhythms) yet described.
Conclusion: These results establish the leech as a new model system for studying aspects of the neuronal basis of reproductive behavior.
2010 Elsevier Ltd. All rights reserved.
Figures


Comment in
-
Behavioral neurobiology: leech lust in the lab.Curr Biol. 2010 Mar 23;20(6):R276-8. doi: 10.1016/j.cub.2010.02.013. Curr Biol. 2010. PMID: 20334835 No abstract available.
Similar articles
-
Possible functions of oxytocin/vasopressin-superfamily peptides in annelids with special reference to reproduction and osmoregulation.J Exp Zool. 1999 Sep 1;284(4):401-6. doi: 10.1002/(sici)1097-010x(19990901)284:4<401::aid-jez6>3.3.co;2-l. J Exp Zool. 1999. PMID: 10451417
-
Annetocin, an annelid oxytocin-related peptide, induces egg-laying behavior in the earthworm, Eisenia foetida.J Exp Zool. 1996 Oct 1;276(2):151-6. doi: 10.1002/(SICI)1097-010X(19961001)276:2<151::AID-JEZ8>3.0.CO;2-N. J Exp Zool. 1996. PMID: 8900077
-
Regulation of Na(+) transport across leech skin by peptide hormones and neurotransmitters.J Exp Biol. 2001 Apr;204(Pt 8):1509-17. doi: 10.1242/jeb.204.8.1509. J Exp Biol. 2001. PMID: 11273812
-
A classic model animal in the 21st century: recent lessons from the leech nervous system.J Exp Biol. 2015 Nov;218(Pt 21):3353-9. doi: 10.1242/jeb.113860. J Exp Biol. 2015. PMID: 26538172 Review.
-
Neural control of heartbeat in the leech and in some other invertebrates.Physiol Rev. 1979 Jan;59(1):101-36. doi: 10.1152/physrev.1979.59.1.101. Physiol Rev. 1979. PMID: 220645 Review.
Cited by
-
Global view of the evolution and diversity of metazoan neuropeptide signaling.Proc Natl Acad Sci U S A. 2013 May 21;110(21):8702-7. doi: 10.1073/pnas.1221833110. Epub 2013 May 1. Proc Natl Acad Sci U S A. 2013. PMID: 23637342 Free PMC article.
-
Oxytocin Involvement in Body Composition Unveils the True Identity of Oxytocin.Int J Mol Sci. 2021 Jun 15;22(12):6383. doi: 10.3390/ijms22126383. Int J Mol Sci. 2021. PMID: 34203705 Free PMC article. Review.
-
Autism, oxytocin and interoception.Neurosci Biobehav Rev. 2014 Nov;47:410-30. doi: 10.1016/j.neubiorev.2014.09.012. Epub 2014 Sep 30. Neurosci Biobehav Rev. 2014. PMID: 25277283 Free PMC article. Review.
-
Expression of arginine vasotocin receptors in the developing zebrafish CNS.Gene Expr Patterns. 2013 Dec;13(8):335-42. doi: 10.1016/j.gep.2013.06.005. Epub 2013 Jul 2. Gene Expr Patterns. 2013. PMID: 23830982 Free PMC article.
-
Responses to conflicting stimuli in a simple stimulus-response pathway.J Neurosci. 2015 Feb 11;35(6):2398-406. doi: 10.1523/JNEUROSCI.3823-14.2015. J Neurosci. 2015. PMID: 25673834 Free PMC article.
References
-
- Insel TR, Young L, Wang Z. Central oxytocin and reproductive behaviours. Rev Reprod. 1997;2:28–37. - PubMed
-
- Ball GF, Balthazar J. Ethological concepts revisited: immediate early gene induction in response to sexual stimuli in birds. Brain Behav Evol. 2001;57:252–270. - PubMed
-
- Greenspan RJ, Ferveur JF. Courtship in Drosophila. Annu Rev Genet. 2000;34:205–232. - PubMed
-
- Fisher H, Aron A, Brown LL. Romantic love: an fMRI study of a neural mechanism for mate choice. J Comp Neurol. 2005;493:58–62. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

