Partitioning of homologous nicotinic acid ester prodrugs (nicotinates) into dipalmitoylphosphatidylcholine (DPPC) membrane bilayers
- PMID: 20227859
- PMCID: PMC2873030
- DOI: 10.1016/j.colsurfb.2010.02.015
Partitioning of homologous nicotinic acid ester prodrugs (nicotinates) into dipalmitoylphosphatidylcholine (DPPC) membrane bilayers
Abstract
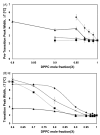
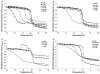
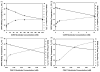
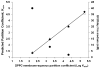
The partitioning behavior of a series of perhydrocarbon nicotinic acid esters (nicotinates) between aqueous solution and dipalmitoylphosphatidylcholine (DPPC) membrane bilayers is investigated as a function of increasing alkyl chain length. The hydrocarbon nicotinates represent putative prodrugs, derivatives of the polar drug nicotinic acid, whose functionalization provides the hydrophobic character necessary for pulmonary delivery in a hydrophobic, fluorocarbon solvent, such as perfluorooctyl bromide. Independent techniques of differential scanning calorimetry and 1,6-diphenyl-1,3,5-hexatriene (DPH) fluorescence anisotropy measurements are used to analyze the thermotropic phase behavior and lipid bilayer fluidity as a function of nicotinate concentration. At increasing concentrations of nicotinates over the DPPC mole fraction range examined (X(DPPC)=0.6-1.0), all the nicotinates (ethyl (C2H5); butyl (C4H9); hexyl (C6H13); and octyl (C8H17)) partition into the lipid bilayer at sufficient levels to eliminate the pretransition, and decrease and broaden the gel to fluid phase transition temperature. The concentration at which these effects occur is chain length-dependent; the shortest chain nicotinate, C2H5, elicits the least dramatic response. Similarly, the DPH anisotropy results demonstrate an alteration of the bilayer organization in the liposomes as a consequence of the chain length-dependent partitioning of the nicotinates into DPPC bilayers. The membrane partition coefficients (logarithm values), determined from the depressed bilayer phase transition temperatures, increase from 2.18 for C2H5 to 5.25 for C8H17. The DPPC membrane/water partitioning of the perhydrocarbon nicotinate series correlates with trends in the octanol/water partitioning of these solutes, suggesting that their incorporation into the bilayer is driven by increasing hydrophobicity.
Copyright 2010 Elsevier B.V. All rights reserved.
Figures


Similar articles
-
A DSC and FTIR spectroscopic study of the effects of the epimeric 4-cholesten-3-ols and 4-cholesten-3-one on the thermotropic phase behaviour and organization of dipalmitoylphosphatidylcholine bilayer membranes: comparison with their 5-cholesten analogues.Chem Phys Lipids. 2014 Jan;177:71-90. doi: 10.1016/j.chemphyslip.2013.11.008. Epub 2013 Dec 1. Chem Phys Lipids. 2014. PMID: 24296232
-
A DSC and FTIR spectroscopic study of the effects of the epimeric cholestan-3-ols and cholestan-3-one on the thermotropic phase behavior and organization of dipalmitoylphosphatidylcholine bilayer membranes: Comparison with their 5-cholesten analogs.Chem Phys Lipids. 2015 Apr;187:34-49. doi: 10.1016/j.chemphyslip.2015.02.002. Epub 2015 Feb 27. Chem Phys Lipids. 2015. PMID: 25732198
-
Interaction of long-chain nicotinates with dipalmitoylphosphatidylcholine.J Lipid Res. 2005 Mar;46(3):535-46. doi: 10.1194/jlr.M400406-JLR200. Epub 2004 Dec 16. J Lipid Res. 2005. PMID: 15604517
-
A calorimetric and spectroscopic comparison of the effects of cholesterol and its immediate biosynthetic precursors 7-dehydrocholesterol and desmosterol on the thermotropic phase behavior and organization of dipalmitoylphosphatidylcholine bilayer membranes.Chem Phys Lipids. 2015 Oct;191:123-35. doi: 10.1016/j.chemphyslip.2015.09.002. Epub 2015 Sep 11. Chem Phys Lipids. 2015. PMID: 26368000
-
A calorimetric and spectroscopic comparison of the effects of lathosterol and cholesterol on the thermotropic phase behavior and organization of dipalmitoylphosphatidylcholine bilayer membranes.Biochemistry. 2011 Nov 22;50(46):9982-97. doi: 10.1021/bi200721j. Epub 2011 Oct 26. Biochemistry. 2011. PMID: 21951051
Cited by
-
Modification of Liposomal Properties by an Engineered Gemini Surfactant.Langmuir. 2025 Feb 11;41(5):3042-3052. doi: 10.1021/acs.langmuir.4c03043. Epub 2025 Jan 25. Langmuir. 2025. PMID: 39862190 Free PMC article.
References
-
- Rongvaux A, Andris F, Van Gool F, Leo O. Bioessays. 2003;25:683. - PubMed
-
- Wang Q, Giri DM, Hyde M, Nakashima JM, Javadi I. J Biochem Toxicol. 1990;5:13. - PubMed
-
- Nagai A, Yasui S, Ozawa Y, Uno H, Konno K. Eur Respir J. 1994;7:1125. - PubMed
-
- Altschul R, Hoffer A, Stephen JD. Arch Biochem Biophys. 1955;54:558. - PubMed
-
- Kamanna VS, Kashyab ML. Curr Atheroscl Rep. 2000;2:36. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

