Co-regulation of caveolar and Cdc42-dependent fluid phase endocytosis by phosphocaveolin-1
- PMID: 20228056
- PMCID: PMC2865320
- DOI: 10.1074/jbc.M109.069427
Co-regulation of caveolar and Cdc42-dependent fluid phase endocytosis by phosphocaveolin-1
Abstract
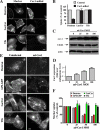
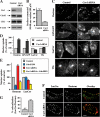
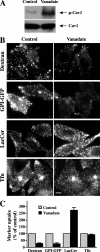
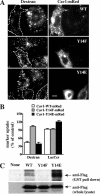
Several clathrin-independent endocytosis mechanisms have been identified that can be distinguished by specific requirements for certain proteins, such as caveolin-1 (Cav1) and the Rho GTPases, RhoA and Cdc42, as well as by specific cargo. Some endocytic pathways may be co-regulated such that disruption of one pathway leads to the up-regulation of another; however, the underlying mechanisms for this are unclear. Cav1 has been reported to function as a guanine nucleotide dissociation inhibitor (GDI), which inhibits Cdc42 activation. We tested the hypothesis that Cav1 can regulate Cdc42-dependent, fluid phase endocytosis. We demonstrate that Cav1 overexpression decreases fluid phase endocytosis, whereas silencing of Cav1 enhances this pathway. Enhancement of Cav1 phosphorylation using a phosphatase inhibitor reduces Cdc42-regulated pinocytosis while stimulating caveolar endocytosis. Fluid phase endocytosis was inhibited by expression of a putative phosphomimetic mutant, Cav1-Y14E, but not by the phospho-deficient mutant, Cav1-Y14F. Overexpression of Cav2, or a Cav1 mutant in which the GDI region was altered to the corresponding sequence in Cav2, did not suppress fluid phase endocytosis. These results suggest that the Cav1 expression level and phosphorylation state regulates fluid phase endocytosis via the interaction between the Cav1 GDI region and Cdc42. These data define a novel molecular mechanism for co-regulation of two distinct clathrin-independent endocytic pathways.
Figures
Similar articles
-
Prominin-2 expression increases protrusions, decreases caveolae and inhibits Cdc42 dependent fluid phase endocytosis.Biochem Biophys Res Commun. 2013 May 10;434(3):466-72. doi: 10.1016/j.bbrc.2013.03.097. Epub 2013 Apr 10. Biochem Biophys Res Commun. 2013. PMID: 23583380 Free PMC article.
-
Endocytic crosstalk: cavins, caveolins, and caveolae regulate clathrin-independent endocytosis.PLoS Biol. 2014 Apr 8;12(4):e1001832. doi: 10.1371/journal.pbio.1001832. eCollection 2014 Apr. PLoS Biol. 2014. PMID: 24714042 Free PMC article.
-
Distinct mechanisms of clathrin-independent endocytosis have unique sphingolipid requirements.Mol Biol Cell. 2006 Jul;17(7):3197-210. doi: 10.1091/mbc.e05-12-1101. Epub 2006 May 3. Mol Biol Cell. 2006. PMID: 16672382 Free PMC article.
-
Membrane microdomains, caveolae, and caveolar endocytosis of sphingolipids.Mol Membr Biol. 2006 Jan-Feb;23(1):101-10. doi: 10.1080/09687860500460041. Mol Membr Biol. 2006. PMID: 16611585 Review.
-
Clathrin-independent endocytosis: new insights into caveolae and non-caveolar lipid raft carriers.Biochim Biophys Acta. 2005 Dec 30;1746(3):349-63. doi: 10.1016/j.bbamcr.2005.11.007. Biochim Biophys Acta. 2005. PMID: 16440447 Review.
Cited by
-
Prominin-2 expression increases protrusions, decreases caveolae and inhibits Cdc42 dependent fluid phase endocytosis.Biochem Biophys Res Commun. 2013 May 10;434(3):466-72. doi: 10.1016/j.bbrc.2013.03.097. Epub 2013 Apr 10. Biochem Biophys Res Commun. 2013. PMID: 23583380 Free PMC article.
-
Clathrin-independent endocytosis: an increasing degree of complexity.Histochem Cell Biol. 2018 Aug;150(2):107-118. doi: 10.1007/s00418-018-1678-5. Epub 2018 May 17. Histochem Cell Biol. 2018. PMID: 29774430 Free PMC article. Review.
-
Endocytic crosstalk: cavins, caveolins, and caveolae regulate clathrin-independent endocytosis.PLoS Biol. 2014 Apr 8;12(4):e1001832. doi: 10.1371/journal.pbio.1001832. eCollection 2014 Apr. PLoS Biol. 2014. PMID: 24714042 Free PMC article.
-
NELL2-cdc42 signaling regulates BAF complexes and Ewing sarcoma cell growth.Cell Rep. 2021 Jul 6;36(1):109254. doi: 10.1016/j.celrep.2021.109254. Cell Rep. 2021. PMID: 34233189 Free PMC article.
-
The Protein Toxins Ricin and Shiga Toxin as Tools to Explore Cellular Mechanisms of Internalization and Intracellular Transport.Toxins (Basel). 2021 May 25;13(6):377. doi: 10.3390/toxins13060377. Toxins (Basel). 2021. PMID: 34070659 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

