Interaction between Plasmodium falciparum apical membrane antigen 1 and the rhoptry neck protein complex defines a key step in the erythrocyte invasion process of malaria parasites
- PMID: 20228060
- PMCID: PMC2863225
- DOI: 10.1074/jbc.M109.080770
Interaction between Plasmodium falciparum apical membrane antigen 1 and the rhoptry neck protein complex defines a key step in the erythrocyte invasion process of malaria parasites
Abstract
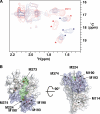
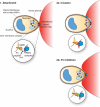
Invasion of host cells by apicomplexan parasites, including Plasmodium falciparum and Toxoplasma gondii, is a multistep process. Central to invasion is the formation of a tight junction, an aperture in the host cell through which the parasite pulls itself before settling into a newly formed parasitophorous vacuole. Two protein groups, derived from different secretory organelles, the micronemal protein AMA1 and the rhoptry proteins RON2, RON4, and RON5, have been shown to form part of this structure, with antibodies targeting P. falciparum AMA1 known to inhibit invasion, probably via disruption of its association with the PfRON proteins. Inhibitory AMA1-binding peptides have also been described that block P. falciparum merozoite invasion of the erythrocyte. One of these, R1, blocks invasion some time after initial attachment to the erythrocyte and reorientation of the merozoite to its apical pole. Here we show that the R1 peptide binds the PfAMA1 hydrophobic trough and demonstrate that binding to this region prevents its interaction with the PfRON complex. We show that this defined association between PfAMA1 and the PfRON complex occurs after reorientation and engagement of the actomyosin motor and argue that it precedes rhoptry release. We propose that the formation of the AMA1-RON complex is essential for secretion of the rhoptry contents, which then allows the establishment of parasite infection within the parasitophorous vacuole.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

