Iowa variant of familial Alzheimer's disease: accumulation of posttranslationally modified AbetaD23N in parenchymal and cerebrovascular amyloid deposits
- PMID: 20228223
- PMCID: PMC2843474
- DOI: 10.2353/ajpath.2010.090636
Iowa variant of familial Alzheimer's disease: accumulation of posttranslationally modified AbetaD23N in parenchymal and cerebrovascular amyloid deposits
Abstract
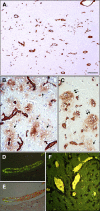
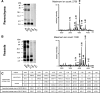
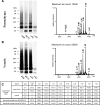
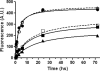
Mutations within the amyloid-beta (Abeta) sequence, especially those clustered at residues 21-23, which are linked to early onset familial Alzheimer's disease (AD), are primarily associated with cerebral amyloid angiopathy (CAA). The basis for this predominant vascular amyloid burden and the differential clinical phenotypes of cerebral hemorrhage/stroke in some patients and dementia in others remain unknown. The AbetaD23N Iowa mutation is associated with progressive AD-like dementia, often without clinically manifested intracerebral hemorrhage. Neuropathologically, the disease is characterized by predominant preamyloid deposits, severe CAA, and abundant neurofibrillary tangles in the presence of remarkably few mature plaques. Biochemical analyses using a combination of immunoprecipitation, mass spectrometry, amino acid sequence, and Western blot analysis performed after sequential tissue extractions to separately isolate soluble components, preamyloid, and fibrillar amyloid species indicated that the Iowa deposits are complex mixtures of mutated and nonmutated Abeta molecules. These molecules exhibited various degrees of solubility, were highly heterogeneous at both the N- and C-termini, and showed partial aspartate isomerization at positions 1, 7, and 23. This collection of Abeta species-the Iowa brain Abeta peptidome-contained clear imprints of amyloid clearance mechanisms yet highlighted the unique neuropathological features shared by a non-Abeta cerebral amyloidosis, familial Danish dementia, in which neurofibrillary tangles coexist with extensive pre-amyloid deposition in the virtual absence of fibrillar lesions. These data therefore challenge the importance of neuritic plaques as the sole contributors for the development of dementia.
Figures


Similar articles
-
Chemical traits of cerebral amyloid angiopathy in familial British-, Danish-, and non-Alzheimer's dementias.J Neurochem. 2022 Nov;163(3):233-246. doi: 10.1111/jnc.15694. Epub 2022 Sep 25. J Neurochem. 2022. PMID: 36102248 Free PMC article.
-
Preamyloid lesions and cerebrovascular deposits in the mechanism of dementia: lessons from non-beta-amyloid cerebral amyloidosis.Neurodegener Dis. 2008;5(3-4):173-5. doi: 10.1159/000113694. Epub 2008 Mar 6. Neurodegener Dis. 2008. PMID: 18322382 Free PMC article. Review.
-
Familial Danish dementia: co-existence of Danish and Alzheimer amyloid subunits (ADan AND A{beta}) in the absence of compact plaques.J Biol Chem. 2005 Nov 4;280(44):36883-94. doi: 10.1074/jbc.M504038200. Epub 2005 Aug 9. J Biol Chem. 2005. PMID: 16091362
-
Differential deposition of amyloid beta peptides in cerebral amyloid angiopathy associated with Alzheimer's disease and vascular dementia.Acta Neuropathol. 2006 May;111(5):430-5. doi: 10.1007/s00401-006-0054-z. Epub 2006 Mar 23. Acta Neuropathol. 2006. PMID: 16555084
-
Alzheimer's disease.Subcell Biochem. 2012;65:329-52. doi: 10.1007/978-94-007-5416-4_14. Subcell Biochem. 2012. PMID: 23225010 Review.
Cited by
-
Matrix metalloproteinase 2 (MMP-2) degrades soluble vasculotropic amyloid-beta E22Q and L34V mutants, delaying their toxicity for human brain microvascular endothelial cells.J Biol Chem. 2010 Aug 27;285(35):27144-27158. doi: 10.1074/jbc.M110.135228. Epub 2010 Jun 24. J Biol Chem. 2010. PMID: 20576603 Free PMC article.
-
Aβ truncated species: Implications for brain clearance mechanisms and amyloid plaque deposition.Biochim Biophys Acta Mol Basis Dis. 2018 Jan;1864(1):208-225. doi: 10.1016/j.bbadis.2017.07.005. Epub 2017 Jul 13. Biochim Biophys Acta Mol Basis Dis. 2018. PMID: 28711595 Free PMC article.
-
Implications of Metal Binding and Asparagine Deamidation for Amyloid Formation.Int J Mol Sci. 2018 Aug 19;19(8):2449. doi: 10.3390/ijms19082449. Int J Mol Sci. 2018. PMID: 30126231 Free PMC article. Review.
-
Amyloidosis associated with cerebral amyloid angiopathy: cell signaling pathways elicited in cerebral endothelial cells.J Alzheimers Dis. 2014;42 Suppl 3(0 3):S167-76. doi: 10.3233/JAD-140027. J Alzheimers Dis. 2014. PMID: 24670400 Free PMC article. Review.
-
In vivo Differential Brain Clearance and Catabolism of Monomeric and Oligomeric Alzheimer's Aβ protein.Front Aging Neurosci. 2016 Sep 27;8:223. doi: 10.3389/fnagi.2016.00223. eCollection 2016. Front Aging Neurosci. 2016. PMID: 27729857 Free PMC article.
References
-
- Ghiso J, Frangione B. Amyloidosis and Alzheimer’s disease. Adv Drug Delivery Rev. 2002;54:1539–1551. - PubMed
-
- Di Fede G, Catania M, Morbin M, Rossi G, Suardi S, Mazzoleni G, Merlin M, Giovagnoli AR, Prioni S, Erbetta A, Falcone C, Gobbi M, Colombo L, Bastone A, Beeg M, Manzoni C, Francescucci B, Spagnoli A, Cantù L, Del Favero E, Levy E, Salmona M, Tagliavini F. A recessive mutation in the APP gene with dominant-negative effect on amyloidogenesis. Science. 2009;323:1473–1477. - PMC - PubMed
-
- Janssen JC, Beck JA, Campbell TA, Dickinson A, Fox NC, Harvey RJ, Houlden H, Rossor MN, Collinge J. Early onset familial Alzheimer’s disease: mutation frequency in 31 families. Neurology. 2003;60:235–239. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

