Do androgens play a beneficial role in the regulation of vascular tone? Nongenomic vascular effects of testosterone metabolites
- PMID: 20228257
- PMCID: PMC2867446
- DOI: 10.1152/ajpheart.00753.2009
Do androgens play a beneficial role in the regulation of vascular tone? Nongenomic vascular effects of testosterone metabolites
Abstract
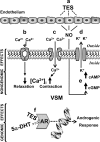
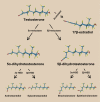
The marked sexual dimorphism that exists in human cardiovascular diseases has led to the dogmatic concept that testosterone (Tes) has deleterious effects and exacerbates the development of cardiovascular disease in males. While some animal studies suggest that Tes does exert deleterious effects by enhancing vascular tone through acute or chronic mechanisms, accumulating evidence suggests that Tes and other androgens exert beneficial effects by inducing rapid vasorelaxation of vascular smooth muscle through nongenomic mechanisms. While this effect frequently has been observed in large arteries at micromolar concentrations, more recent studies have reported vasorelaxation of smaller resistance arteries at nanomolar (physiological) concentrations. The key mechanism underlying Tes-induced vasorelaxation appears to be the modulation of vascular smooth muscle ion channel function, particularly the inactivation of L-type voltage-operated Ca(2+) channels and/or the activation of voltage-operated and Ca(2+)-activated K(+) channels. Studies employing Tes analogs and metabolites reveal that androgen-induced vasodilation is a structurally specific nongenomic effect that is fundamentally different than the genomic effects on reproductive targets. For example, 5alpha-dihydrotestosterone exhibits potent genomic-androgenic effects but only moderate vasorelaxing activity, whereas its isomer 5beta-dihydrotestosterone is devoid of androgenic effects but is a highly efficacious vasodilator. These findings suggest that the dihydro-metabolites of Tes or other androgen analogs devoid of androgenic or estrogenic effects could have useful therapeutic roles in hypertension, erectile dysfunction, prostatic ischemia, or other vascular dysfunctions.
Figures
Similar articles
-
Testosterone-induced relaxation of rat aorta is androgen structure specific and involves K+ channel activation.J Appl Physiol (1985). 2001 Dec;91(6):2742-50. doi: 10.1152/jappl.2001.91.6.2742. J Appl Physiol (1985). 2001. PMID: 11717242
-
Relaxation of androgens on rat thoracic aorta: testosterone concentration dependent agonist/antagonist L-type Ca2+ channel activity, and 5beta-dihydrotestosterone restricted to L-type Ca2+ channel blockade.Endocrinology. 2008 May;149(5):2517-26. doi: 10.1210/en.2007-1288. Epub 2008 Feb 14. Endocrinology. 2008. PMID: 18276759
-
Antihypertensive effects of androgens in conscious, spontaneously hypertensive rats.J Steroid Biochem Mol Biol. 2017 Mar;167:106-114. doi: 10.1016/j.jsbmb.2016.11.016. Epub 2016 Nov 23. J Steroid Biochem Mol Biol. 2017. PMID: 27888135
-
Androgens and Non-Genomic vascular responses in hypertension.Biochem Pharmacol. 2022 Sep;203:115200. doi: 10.1016/j.bcp.2022.115200. Epub 2022 Aug 1. Biochem Pharmacol. 2022. PMID: 35926652 Review.
-
Androgens are effective bronchodilators with anti-inflammatory properties: A potential alternative for asthma therapy.Steroids. 2020 Jan;153:108509. doi: 10.1016/j.steroids.2019.108509. Epub 2019 Oct 3. Steroids. 2020. PMID: 31586608 Review.
Cited by
-
Effect of 5β-dihydrotestosterone on vasodilator function and on cell proliferation.PLoS One. 2024 Oct 23;19(10):e0312080. doi: 10.1371/journal.pone.0312080. eCollection 2024. PLoS One. 2024. PMID: 39441776 Free PMC article.
-
Gonadotropin and sex steroid levels in HIV-infected premenopausal women and their association with subclinical atherosclerosis in HIV-infected and -uninfected women in the women's interagency HIV study (WIHS).J Clin Endocrinol Metab. 2013 Apr;98(4):E610-8. doi: 10.1210/jc.2012-3195. Epub 2013 Feb 15. J Clin Endocrinol Metab. 2013. PMID: 23418313 Free PMC article.
-
Systemic hypotensive effects of testosterone are androgen structure-specific and neuronal nitric oxide synthase-dependent.Am J Physiol Regul Integr Comp Physiol. 2015 Jul 15;309(2):R189-95. doi: 10.1152/ajpregu.00110.2015. Epub 2015 May 6. Am J Physiol Regul Integr Comp Physiol. 2015. PMID: 25947172 Free PMC article.
-
Reduced estradiol-induced vasodilation and poly-(ADP-ribose) polymerase (PARP) activity in the aortas of rats with experimental polycystic ovary syndrome (PCOS).PLoS One. 2013;8(3):e55589. doi: 10.1371/journal.pone.0055589. Epub 2013 Mar 26. PLoS One. 2013. PMID: 23555555 Free PMC article.
-
Strategies and methods to study sex differences in cardiovascular structure and function: a guide for basic scientists.Biol Sex Differ. 2011 Dec 12;2:14. doi: 10.1186/2042-6410-2-14. Biol Sex Differ. 2011. PMID: 22152231 Free PMC article.
References
-
- Benagiano G, Mancuso S, Mancuso FP, Wiqvist N, Diczfalusy E. Studies on the metabolism of C-19 steroids in the human foeto-placental unit. 3. Dehydrogenation and reduction products formed by previable foetuses with androstenedione and testosterone. Acta Endocrinol (Copenh) 57: 187–207, 1968 - PubMed
-
- Cairraö E, Alvarez E, Santos-Silva AJ, Verde I. Potassium channels are involved in testosterone-induced vasorelaxation of human umbilical artery. Naunyn Schmiedebergs Arch Pharmacol 376: 375–383, 2008 - PubMed
-
- Chou TM, Sudhir K, Hutchison SJ, Ko E, Amidon TM, Collins P, Chatterjee K. Testosterone induces dilation of canine coronary conductance and resistance arteries in vivo. Circulation 94: 2614–2619, 1996 - PubMed
-
- Corona G, Mannucci E, Lotti F, Fisher AD, Bandini E, Balercia G, Forti G, Maggi M. Pulse pressure, an index of arterial stiffness, is associated with androgen deficiency and impaired penile blood flow in men with ED. J Sex Med 6: 285–293, 2009 - PubMed
-
- Costarella CE, Stallone JN, Rutecki GW, Whittier FC. Testosterone causes direct relaxation of rat thoracic aorta. J Pharmacol Exp Ther 277: 34–39, 1996 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

