GPG-NH2 acts via the metabolite alphaHGA to target HIV-1 Env to the ER-associated protein degradation pathway
- PMID: 20230608
- PMCID: PMC2846875
- DOI: 10.1186/1742-4690-7-20
GPG-NH2 acts via the metabolite alphaHGA to target HIV-1 Env to the ER-associated protein degradation pathway
Abstract
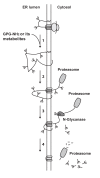
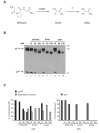
Background: The synthetic peptide glycyl-prolyl-glycine amide (GPG-NH2) was previously shown to abolish the ability of HIV-1 particles to fuse with the target cells, by reducing the content of the viral envelope glycoprotein (Env) in progeny HIV-1 particles. The loss of Env was found to result from GPG-NH2 targeting the Env precursor protein gp160 to the ER-associated protein degradation (ERAD) pathway during its maturation. However, the anti-viral effect of GPG-NH2 has been shown to be mediated by its metabolite alpha-hydroxy-glycineamide (alphaHGA), which is produced in the presence of fetal bovine serum, but not human serum. In accordance, we wanted to investigate whether the targeting of gp160 to the ERAD pathway by GPG-NH2 was attributed to its metabolite alphaHGA.
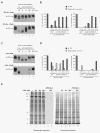
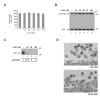
Results: In the presence of fetal bovine serum, GPG-NH2, its intermediary metabolite glycine amide (G-NH2), and final metabolite alphaHGA all induced the degradation of gp160 through the ERAD pathway. However, when fetal bovine serum was replaced with human serum only alphaHGA showed an effect on gp160, and this activity was further shown to be completely independent of serum. This indicated that GPG-NH2 acts as a pro-drug, which was supported by the observation that it had to be added earlier to the cell cultures than alphaHGA to induce the degradation of gp160. Furthermore, the substantial reduction of Env incorporation into HIV-1 particles that occurs during GPG-NH2 treatment was also achieved by treating HIV-1 infected cells with alphaHGA.
Conclusions: The previously observed specificity of GPG-NH2 towards gp160 in HIV-1 infected cells, resulting in the production of Env (gp120/gp41) deficient fusion incompetent HIV-1 particles, was most probably due to the action of the GPG-NH2 metabolite alphaHGA.
Figures

Similar articles
-
Anti-human immunodeficiency virus type 1 agent alpha-hydroxy glycineamide enters the target cells via a mechanism of passive diffusion.J Pharm Pharmacol. 2014 Oct;66(10):1388-93. doi: 10.1111/jphp.12269. Epub 2014 Apr 29. J Pharm Pharmacol. 2014. PMID: 24780097
-
The tripeptide glycyl-prolyl-glycine amide does not affect the early steps of the human immunodeficiency virus type 1 replication.J Hum Virol. 2001 Jan-Feb;4(1):8-15. J Hum Virol. 2001. PMID: 11213934
-
Small molecule targets Env for endoplasmic reticulum-associated protein degradation and inhibits human immunodeficiency virus type 1 propagation.J Virol. 2009 Oct;83(19):10075-84. doi: 10.1128/JVI.01700-08. Epub 2009 Jul 29. J Virol. 2009. PMID: 19640982 Free PMC article.
-
Obligatory involvement of CD26/dipeptidyl peptidase IV in the activation of the antiretroviral tripeptide glycylprolylglycinamide (GPG-NH(2)).Int J Biochem Cell Biol. 2004 Sep;36(9):1848-59. doi: 10.1016/j.biocel.2004.02.017. Int J Biochem Cell Biol. 2004. PMID: 15183349
-
HIV-1 gp41: mediator of fusion and target for inhibition.AIDS Rev. 2003 Oct-Dec;5(4):214-21. AIDS Rev. 2003. PMID: 15012000 Review.
References
-
- Kilgore NR, Salzwedel K, Reddick M, Allaway GP, Wild CT. Direct evidence that C-peptide inhibitors of human immunodeficiency virus type 1 entry bind to the gp41 N-helical domain in receptor-activated viral envelope. J Virol. 2003;77:7669–7672. doi: 10.1128/JVI.77.13.7669-7672.2003. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

