Variations in the rheostat model of apoptosis: what studies of retinal ganglion cell death tell us about the functions of the Bcl2 family proteins
- PMID: 20230818
- PMCID: PMC2885977
- DOI: 10.1016/j.exer.2010.03.004
Variations in the rheostat model of apoptosis: what studies of retinal ganglion cell death tell us about the functions of the Bcl2 family proteins
Abstract
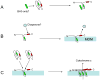
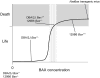
Studies of the functions of members of the Bcl2 gene family suggested that apoptosis was controlled by a rheostat in which anti-apoptotic proteins like BCL2 bound and sequestered pro-apoptotic proteins like BAX. Our current understanding of these proteins suggests that this is a simplistic model. The new rheostat model predicts that BH3-only peptides act as neutralizing ligands for the anti-apoptotic proteins, thus allowing molecules like BAX to become activated and initiate mitochondrial dysfunction - a critical step in the intrinsic apoptotic program. Studies of retinal ganglion cell apoptosis indicate that a threshold of BAX expression is required for its successful activation, which is independent of the overall concentration of anti-apoptotic proteins in these cells.
Copyright 2010 Elsevier Ltd. All rights reserved.
Figures

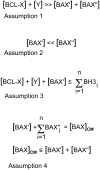
Similar articles
-
BAX to basics: How the BCL2 gene family controls the death of retinal ganglion cells.Prog Retin Eye Res. 2017 Mar;57:1-25. doi: 10.1016/j.preteyeres.2017.01.002. Epub 2017 Jan 4. Prog Retin Eye Res. 2017. PMID: 28064040 Free PMC article. Review.
-
Involvement of the Bcl2 gene family in the signaling and control of retinal ganglion cell death.Prog Brain Res. 2008;173:423-35. doi: 10.1016/S0079-6123(08)01129-1. Prog Brain Res. 2008. PMID: 18929125 Review.
-
Interaction of E1B 19K with Bax is required to block Bax-induced loss of mitochondrial membrane potential and apoptosis.Oncogene. 1998 Dec 10;17(23):2993-3005. doi: 10.1038/sj.onc.1202215. Oncogene. 1998. PMID: 9881701
-
Up-regulation of Bax protein in degenerating retinal ganglion cells precedes apoptotic cell death after optic nerve lesion in the rat.Eur J Neurosci. 1997 Aug;9(8):1763-72. doi: 10.1111/j.1460-9568.1997.tb01534.x. Eur J Neurosci. 1997. PMID: 9283831
-
Rat retinal ganglion cells upregulate the pro-apoptotic BH3-only protein Bim after optic nerve transection.Brain Res Mol Brain Res. 2003 Dec 12;120(1):30-7. doi: 10.1016/j.molbrainres.2003.09.016. Brain Res Mol Brain Res. 2003. PMID: 14667574
Cited by
-
A proteomics view of the molecular mechanisms and biomarkers of glaucomatous neurodegeneration.Prog Retin Eye Res. 2013 Jul;35:18-43. doi: 10.1016/j.preteyeres.2013.01.004. Epub 2013 Feb 5. Prog Retin Eye Res. 2013. PMID: 23396249 Free PMC article. Review.
-
Neurodegenerative and inflammatory pathway components linked to TNF-α/TNFR1 signaling in the glaucomatous human retina.Invest Ophthalmol Vis Sci. 2011 Oct 31;52(11):8442-54. doi: 10.1167/iovs.11-8152. Invest Ophthalmol Vis Sci. 2011. PMID: 21917936 Free PMC article.
-
BAX-Depleted Retinal Ganglion Cells Survive and Become Quiescent Following Optic Nerve Damage.Mol Neurobiol. 2020 Feb;57(2):1070-1084. doi: 10.1007/s12035-019-01783-7. Epub 2019 Oct 31. Mol Neurobiol. 2020. PMID: 31673950 Free PMC article.
-
BBC3 (PUMA) regulates developmental apoptosis but not axonal injury induced death in the retina.Mol Neurodegener. 2011 Jul 15;6:50. doi: 10.1186/1750-1326-6-50. Mol Neurodegener. 2011. PMID: 21762490 Free PMC article.
-
Simvastatin inhibits ischemia/reperfusion injury-induced apoptosis of retinal cells via downregulation of the tumor necrosis factor-α/nuclear factor-κB pathway.Int J Mol Med. 2015 Aug;36(2):399-405. doi: 10.3892/ijmm.2015.2244. Epub 2015 Jun 11. Int J Mol Med. 2015. PMID: 26063345 Free PMC article.
References
-
- Adams JM, Cory S. The bcl-2 protein family: arbiters of cell survival. Science. 1998;281:1322–1326. - PubMed
-
- Antonsson B, Conti F, Ciavatta A, Montessuit S, Lewis S, Martinou I, Bernasconi L, Bernard A, Mermod JJ, Mazzei G, Maundrell K, Gambale F, Sadoul R, Martinou JC. Inhibition of Bax channel-forming activity by Bcl-2. Science. 1997;277:370–372. - PubMed
-
- Antonsson B, Montessuit S, Sanchez B, Martinou JC. Bax is present as a high molecular weight oligomer/complex in the mitochondrial membrane of apoptotic cells. J Biol Chem. 2001;276:11615–11623. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials


