Acute modulation of sugar transport in brain capillary endothelial cell cultures during activation of the metabolic stress pathway
- PMID: 20231288
- PMCID: PMC2865294
- DOI: 10.1074/jbc.M110.110593
Acute modulation of sugar transport in brain capillary endothelial cell cultures during activation of the metabolic stress pathway
Abstract
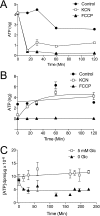
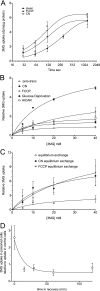
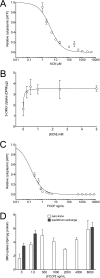
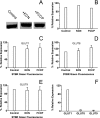
GLUT1-catalyzed equilibrative sugar transport across the mammalian blood-brain barrier is stimulated during acute and chronic metabolic stress; however, the mechanism of acute transport regulation is unknown. We have examined acute sugar transport regulation in the murine brain microvasculature endothelial cell line bEnd.3. Acute cellular metabolic stress was induced by glucose depletion, by potassium cyanide, or by carbonyl cyanide p-trifluoromethoxyphenylhydrazone, which reduce or deplete intracellular ATP within 15 min. This results in a 1.7-7-fold increase in V(max) for zero-trans 3-O-methylglucose uptake (sugar uptake into sugar-free cells) and a 3-10-fold increase in V(max) for equilibrium exchange transport (intracellular [sugar] = extracellular [sugar]). GLUT1, GLUT8, and GLUT9 mRNAs are detected in bEnd.3 cells where GLUT1 mRNA levels are 33-fold greater than levels of GLUT8 or GLUT9 mRNA. Neither GLUT1 mRNA nor total protein levels are affected by acute metabolic stress. Cell surface biotinylation reveals that plasma membrane GLUT1 levels are increased 2-3-fold by metabolic depletion, although cell surface Na(+),K(+)-ATPase levels remain unaffected by ATP depletion. Treatment with the AMP-activated kinase agonist, AICAR, increases V(max) for net 3-O-methylglucose uptake by 2-fold. Glucose depletion and treatment with potassium cyanide, carbonyl cyanide p-trifluoromethoxyphenylhydrazone, and AICAR also increase AMP-dependent kinase phosphorylation in bEnd.3 cells. These results suggest that metabolic stress rapidly stimulates blood-brain barrier endothelial cell sugar transport by acute up-regulation of plasma membrane GLUT1 levels, possibly involving AMP-activated kinase activity.
Figures

Similar articles
-
AMP kinase regulation of sugar transport in brain capillary endothelial cells during acute metabolic stress.Am J Physiol Cell Physiol. 2012 Oct 15;303(8):C806-14. doi: 10.1152/ajpcell.00437.2011. Epub 2012 Jul 3. Am J Physiol Cell Physiol. 2012. PMID: 22763120 Free PMC article.
-
Regulation of GLUT1-mediated sugar transport by an antiport/uniport switch mechanism.Biochemistry. 1996 Oct 8;35(40):13231-9. doi: 10.1021/bi961208t. Biochemistry. 1996. PMID: 8855962
-
ATP-dependent sugar transport complexity in human erythrocytes.Am J Physiol Cell Physiol. 2007 Feb;292(2):C974-86. doi: 10.1152/ajpcell.00335.2006. Epub 2006 Aug 23. Am J Physiol Cell Physiol. 2007. PMID: 16928769 Free PMC article.
-
The red blood cell glucose transporter presents multiple, nucleotide-sensitive sugar exit sites.Biochemistry. 2001 Dec 25;40(51):15549-61. doi: 10.1021/bi015586w. Biochemistry. 2001. PMID: 11747430
-
Enhancement of glucose transport by vascular endothelial growth factor in retinal endothelial cells.Invest Ophthalmol Vis Sci. 2000 Jun;41(7):1876-84. Invest Ophthalmol Vis Sci. 2000. PMID: 10845612
Cited by
-
AMP kinase regulation of sugar transport in brain capillary endothelial cells during acute metabolic stress.Am J Physiol Cell Physiol. 2012 Oct 15;303(8):C806-14. doi: 10.1152/ajpcell.00437.2011. Epub 2012 Jul 3. Am J Physiol Cell Physiol. 2012. PMID: 22763120 Free PMC article.
-
Reconciling contradictory findings: Glucose transporter 1 (GLUT1) functions as an oligomer of allosteric, alternating access transporters.J Biol Chem. 2017 Dec 22;292(51):21035-21046. doi: 10.1074/jbc.M117.815589. Epub 2017 Oct 24. J Biol Chem. 2017. PMID: 29066623 Free PMC article.
-
Role of the GLUT1 Glucose Transporter in Postnatal CNS Angiogenesis and Blood-Brain Barrier Integrity.Circ Res. 2020 Jul 31;127(4):466-482. doi: 10.1161/CIRCRESAHA.119.316463. Epub 2020 May 14. Circ Res. 2020. PMID: 32404031 Free PMC article.
-
Glucose Transporters at the Blood-Brain Barrier: Function, Regulation and Gateways for Drug Delivery.Mol Neurobiol. 2017 Mar;54(2):1046-1077. doi: 10.1007/s12035-015-9672-6. Epub 2016 Jan 22. Mol Neurobiol. 2017. PMID: 26801191 Review.
-
PKCs Sweeten Cell Metabolism by Phosphorylation of Glut1.Mol Cell. 2015 Jun 4;58(5):711-2. doi: 10.1016/j.molcel.2015.05.025. Mol Cell. 2015. PMID: 26046643 Free PMC article.
References
-
- Gerhart D. Z., LeVasseur R. J., Broderius M. A., Drewes L. R. (1989) J. Neurosci. Res. 22, 464–472 - PubMed
-
- Kalaria R. N., Gravina S. A., Schmidley J. W., Perry G., Harik S. I. (1988) Ann. Neurol. 24, 757–764 - PubMed
-
- Pardridge W. M., Boado R. J., Farrell C. R. (1990) J. Biol. Chem. 265, 18035–18040 - PubMed
-
- Takata K., Kasahara T., Kasahara M., Ezaki O., Hirano H. (1990) Biochem. Biophys. Res. Commun. 173, 67–73 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

